(R)-[1-(4-Bromophenyl)ethyl]carbamic acid tert-butyl ester
- CAS No.
- 578729-21-2
- Chemical Name:
- (R)-[1-(4-Bromophenyl)ethyl]carbamic acid tert-butyl ester
- Synonyms
- (R)-N-Boc-1-(4-broMophenyl)ethylaMine;tert-butyl (R)-1-(4-bromophenyl)ethylcarbamate;(R)-tert-butyl (1-(4-broMophenyl)ethyl)carbaMate;tert-Butyl [(1R)-1-(4-bromophenyl)ethyl]carbamate;tert-butyl N-[(1R)-1-(4-broMophenyl)ethyl]carbaMate;(R)-[1-(4-Bromophenyl)ethyl]carbamic acid tert-butyl ester;Carbamic acid, N-[(1R)-1-(4-bromophenyl)ethyl]-, 1,1-dimethylethyl ester
- CBNumber:
- CB22128762
- Molecular Formula:
- C13H18BrNO2
- Molecular Weight:
- 300.19
- MDL Number:
- MFCD11506010
- MOL File:
- 578729-21-2.mol
- MSDS File:
- SDS
| Product description | Number | Pack Size | Price |
| (R)-tert-Butyl(1-(4-Bromophenyl)ethyl)carbamate | B789370 | 500mg | $130 |
| (R)-TERT-BUTYL-(1-(4-BROMOPHENYL)ETHYL)CARBAMATE 95.00% | CCH0033069 | 1G | $228.9 |
| (R)-tert-Butyl(1-(4-bromophenyl)ethyl)carbamate | 8549AB | 5g | $447 |
| (R)-tert-Butyl(1-(4-bromophenyl)ethyl)carbamate | 8549AB | 10g | $779 |
| (R)-[1-(4-Bromo-phenyl)-ethyl]-carbamicacidtert-butylester 96% | 20R0551 | 25g | $998 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|---|---|
| Signal word | Warning |
| Hazard statements | H335-H315-H319 |
| Precautionary statements | P264-P280-P302+P352-P321-P332+P313-P362-P264-P280-P305+P351+P338-P337+P313P |
(R)-[1-(4-Bromophenyl)ethyl]carbamic acid tert-butyl ester price More Price(20)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| TRC | B789370 | (R)-tert-Butyl(1-(4-Bromophenyl)ethyl)carbamate | 578729-21-2 | 500mg | $130 | 2021-12-16 | Buy |
| American Custom Chemicals Corporation | CCH0033069 | (R)-TERT-BUTYL-(1-(4-BROMOPHENYL)ETHYL)CARBAMATE 95.00% | 578729-21-2 | 1G | $228.9 | 2021-12-16 | Buy |
| AK Scientific | 8549AB | (R)-tert-Butyl(1-(4-bromophenyl)ethyl)carbamate | 578729-21-2 | 5g | $447 | 2021-12-16 | Buy |
| AK Scientific | 8549AB | (R)-tert-Butyl(1-(4-bromophenyl)ethyl)carbamate | 578729-21-2 | 10g | $779 | 2021-12-16 | Buy |
| J&W Pharmlab | 20R0551 | (R)-[1-(4-Bromo-phenyl)-ethyl]-carbamicacidtert-butylester 96% | 578729-21-2 | 25g | $998 | 2021-12-16 | Buy |
(R)-[1-(4-Bromophenyl)ethyl]carbamic acid tert-butyl ester Chemical Properties,Uses,Production
Uses
[(1R)-1-(4-bromophenyl)ethyl] tert-butyl carbamate is a carboxylic acid ester organic compound that can be used as a pharmaceutical intermediate.
Synthesis
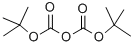
24424-99-5

24358-62-1
![(R)-[1-(4-Bromophenyl)ethyl]carbamic acid tert-butyl ester](/CAS/GIF/578729-21-2.gif)
578729-21-2
General procedure for the synthesis of tert-butyl [(1R)-1-(4-bromophenyl)ethyl]carbamate from di-tert-butyl dicarbonate and 1-(4-bromophenyl)ethylamine: 1. 1-(4-Bromophenyl)ethylamine (1.0 g, 5 mmol) was dissolved in methanol (10 ml) and sodium bicarbonate (1.26 g, 15.0 mmol) and di-tert-butyl dicarbonate (1.2 g, 5.5 mmol) were added. 2. The reaction mixture was sonicated for 4 hours. 3. The solvent was evaporated and the residue partitioned between ethyl acetate and water. 4. The organic phase was washed with brine, dried over anhydrous magnesium sulfate, and the solvent was removed under reduced pressure to afford tert-butyl [(1R)-1-(4-bromophenyl)ethyl]carbamate (1.8 g, 6.0 mmol, 120%) as a white solid. 5. tert-Butyl [(1R)-1-(4-bromophenyl)ethyl]carbamate (0.73 g, 2.4 mmol) was dissolved in 1,2-dimethoxyethane (10 ml), tetrakis(triphenylphosphine)palladium (0.14 g, 0.12 mmol), 2-chloro-pyridinyl-3-boronic acid (0.77 g, 4.9 mmol), and 2 M sodium carbonate solution were added. 6. The reaction mixture was heated to reflux for 16 h. The solvent was removed under reduced pressure. 7. The residue was partitioned between ethyl acetate and water. 8. The organic phase was washed with brine, dried over anhydrous magnesium sulfate and the solvent was removed under reduced pressure. 9. The residue was separated by chromatography on silica gel, eluting with 3:1 heptane/ethyl acetate, and the solvent was removed under reduced pressure to afford tert-butyl (1R)-1-[4-(2-chloropyridin-3-yl)phenyl]ethylcarbamate (0.56 g, 1.7 mmol, 71%) as an off-white solid. 10. tert-Butyl (1R)-1-[4-(2-chloropyridin-3-yl)phenyl]ethylcarbamate (100 mg, 0.3 mmol), sodium ethoxide (61 mg, 0.9 mmol) and tetrahydrofuran (5 ml) were heated and refluxed under argon atmosphere for 16 hours. 11. The solvent was evaporated and the residue was purified on a SCX column eluting with a methanol solution of 2M ammonia to give (1R)-1-[4-(2-ethoxypyridin-3-yl)phenyl]ethylamine (66 mg, 0.27 mmol, 90%) as a yellow gel. 12. (1R)-1-[4-(2-ethoxypyridin-3-yl)phenyl]ethylamine (13 mg, 0.05 mmol) was dissolved in dichloromethane (1 ml), triethylamine (17 mg, 0.16 mmol) and 2-(trifluoromethoxy)benzenesulfonyl chloride (17 mg, 0.065 mmol) were added. 13. The reaction mixture was stirred for 16 hours and the solvent was evaporated under reduced pressure. 14. The crude product was dissolved in dimethyl sulfoxide (1 ml) and purified by preparative LCMS. 15. The solvent was evaporated under reduced pressure to give the title compound (6 mg, 0.013 mmol, 26%). 1H NMR (400MHz, DMSO-d6): δ 8.42 (d, 1H), 8.13 (dd, 1H), 7.81 (dd, 1H), 7.63 (m, 2H), 7.39 (m, 4H), 7.24 (d, 2H), 7.05 (m, 1H), 4.48 (q, 1H), 4.36 (q, 2H), 1.35 (d , 3H), 1.29 (t, 3H) ppm; MS (ESI) m/z: 467 [M + H]+.
References
[1] Patent: US2007/149577, 2007, A1. Location in patent: Page/Page column 6-7
[2] European Journal of Medicinal Chemistry, 2019, p. 407 - 422
[3] Patent: WO2006/18725, 2006, A1. Location in patent: Page/Page column 176
[4] Patent: WO2008/68507, 2008, A2. Location in patent: Page/Page column 57
[5] Patent: WO2009/60278, 2009, A1. Location in patent: Page/Page column 48
(R)-[1-(4-Bromophenyl)ethyl]carbamic acid tert-butyl ester Preparation Products And Raw materials
Raw materials
Preparation Products
(R)-[1-(4-Bromophenyl)ethyl]carbamic acid tert-butyl ester Suppliers
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| HaBo Hong Kong Co., Limited. | +86-25-18512596065 | info@habotech.com | CHINA | 933 | 58 |
| Accela ChemBio Inc. | +1-858-6993322 | info@accelachem.com | United States | 21193 | 58 |
| BOC Sciences | +1-631-485-4226 | inquiry@bocsci.com | United States | 19552 | 58 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | factory@coreychem.com | China | 29795 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-29-87569266 +86-17392581500 | 1015@dideu.com | China | 3992 | 58 |
| Nextpeptide Inc | +86-0571-81612335 +8613336028439 | sales@nextpeptide.com | China | 19908 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418684 +8618949823763 | sales@tnjchem.com | China | 25356 | 58 |
| Suzhou ARTK Medchem Co., Ltd. | +8618118463618 | sales1@artkmedchem.com | China | 39020 | 58 |
| Amadis Chemical Company Limited | 571-89925085 | sales@amadischem.com | China | 131957 | 58 |
| GL Biochem (Shanghai) Ltd | 21-61263452 13641803416 | ymbetter@glbiochem.com | China | 9981 | 64 |
View Lastest Price from (R)-[1-(4-Bromophenyl)ethyl]carbamic acid tert-butyl ester manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
![(R)-[1-(4-Bromophenyl)ethyl]carbamic acid tert-butyl ester pictures](https://img.chemicalbook.com/ProductImageEN/2019-12/Small/e1bc5712-3646-425c-8883-4b18033bde77.png) |
2019-12-26 | (R)-[1-(4-Bromophenyl)ethyl]carbamic acid tert-butyl ester
578729-21-2
|
US $2.00 / KG | 1KG | 98%MIN | ASK | Career Henan Chemical Co |
-
![(R)-[1-(4-Bromophenyl)ethyl]carbamic acid tert-butyl ester pictures](https://img.chemicalbook.com/ProductImageEN/2019-12/Small/e1bc5712-3646-425c-8883-4b18033bde77.png)
- (R)-[1-(4-Bromophenyl)ethyl]carbamic acid tert-butyl ester
578729-21-2
- US $2.00 / KG
- 98%MIN
- Career Henan Chemical Co
578729-21-2((R)-[1-(4-Bromophenyl)ethyl]carbamic acid tert-butyl ester)Related Search:
1of4





![(R)-[1-(4-Bromophenyl)ethyl]carbamic acid tert-butyl ester Structure](CAS/GIF/578729-21-2.gif)