6-METHOXY-1H-INDOLE-2-CARBOXYLIC ACID
- CAS No.
- 16732-73-3
- Chemical Name:
- 6-METHOXY-1H-INDOLE-2-CARBOXYLIC ACID
- Synonyms
- 6-METHOXYINDOLE-2-CARBOXYLIC ACID;NSC 27988;AKOS JY2082559;6-Methoxindole-2-Carboxylic Acid;6-Methoxy-1H-indole-2-carboxylic aci;6-Methoxy-1H-indol-2-carboxylic acid;6-Methoxyindole-2-carboxylicacid,95%;Indole-2-carboxylic acid, 6-methoxy-;6-Methoxyindole-2-carboxylic acid 95%;6-METHOXY-1H-INDOLE-2-CARBOXYLIC ACID
- CBNumber:
- CB2341464
- Molecular Formula:
- C10H9NO3
- Molecular Weight:
- 191.18
- MDL Number:
- MFCD01548823
- MOL File:
- 16732-73-3.mol
- MSDS File:
- SDS
| Product description | Number | Pack Size | Price |
| 6-Methoxyindole-2-carboxylic acid 95% | 722014 | 1g | $46.5 |
| 6-Methoxyindole-2-carboxylic acid | M262800 | 2.5g | $110 |
| 6-Methoxyindole-2-carboxylic acid | J52566 | 5g | $49 |
| 6-Methoxy-1H-indole-2-carboxylic acid | 012222 | 500mg | $10 |
| 6-Methoxy-1H-indole-2-carboxylic acid | 012222 | 1g | $15 |
| More product size | |||
| Melting point | 198-203℃ |
|---|---|
| Boiling point | 447.6±25.0 °C(Predicted) |
| Density | 1.381±0.06 g/cm3(Predicted) |
| storage temp. | Sealed in dry,Room Temperature |
| solubility | DMSO (Slightly) |
| pka | 4.54±0.30(Predicted) |
| form | Solid |
| color | Light Pink |
| InChI | 1S/C10H9NO3/c1-14-7-3-2-6-4-9(10(12)13)11-8(6)5-7/h2-5,11H,1H3,(H,12,13) |
| InChIKey | XNBGANWAZJWOHS-UHFFFAOYSA-N |
| SMILES | COc1ccc2cc([nH]c2c1)C(O)=O |
| CAS DataBase Reference | 16732-73-3(CAS DataBase Reference) |
| UNSPSC Code | 12352100 |
| NACRES | NA.22 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H315-H319-H335 | |||||||||
| Precautionary statements | P261-P264-P271-P280-P302+P352-P305+P351+P338 | |||||||||
| target organs | Respiratory system | |||||||||
| Hazard Codes | Xi | |||||||||
| Risk Statements | 36/37/38 | |||||||||
| Safety Statements | 26 | |||||||||
| WGK Germany | 3 | |||||||||
| HazardClass | IRRITANT | |||||||||
| HS Code | 2933998090 | |||||||||
| Storage Class | 11 - Combustible Solids | |||||||||
| Hazard Classifications | Eye Irrit. 2 Skin Irrit. 2 STOT SE 3 |
|||||||||
| NFPA 704 |
|
6-METHOXY-1H-INDOLE-2-CARBOXYLIC ACID price More Price(29)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 722014 | 6-Methoxyindole-2-carboxylic acid 95% | 16732-73-3 | 1g | $46.5 | 2023-06-20 | Buy |
| TRC | M262800 | 6-Methoxyindole-2-carboxylic acid | 16732-73-3 | 2.5g | $110 | 2021-12-16 | Buy |
| AK Scientific | J52566 | 6-Methoxyindole-2-carboxylic acid | 16732-73-3 | 5g | $49 | 2021-12-16 | Buy |
| Matrix Scientific | 012222 | 6-Methoxy-1H-indole-2-carboxylic acid | 16732-73-3 | 500mg | $10 | 2021-12-16 | Buy |
| Matrix Scientific | 012222 | 6-Methoxy-1H-indole-2-carboxylic acid | 16732-73-3 | 1g | $15 | 2021-12-16 | Buy |
6-METHOXY-1H-INDOLE-2-CARBOXYLIC ACID Chemical Properties,Uses,Production
Uses
• ;Reactant for demethylation reactions using ionic liquids under microwave irradiation1• ;Reactant for preparation of pyrazinoindoledione via Ugi reaction and microwave-assisted cyclization2• ;Reactant for preparation of isoquinolinecarboxamides and their derivatives as opioid receptor antagonists3• ;Synthetic intermediate for preparation of indole fatty alcohol derivatives4• ;Reactant for preparation of acylaminoalkylindoles as MT2-selective melatonin antagonists5
Uses
- Reactant for demethylation reactions using ionic liquids under microwave irradiation
- Reactant for preparation of pyrazinoindoledione via Ugi reaction and microwave-assisted cyclization
- Reactant for preparation of isoquinolinecarboxamides and their derivatives as opioid receptor antagonists
- Synthetic intermediate for preparation of indole fatty alcohol derivatives
- Reactant for preparation of acylaminoalkylindoles as MT2-selective melatonin antagonists
Synthesis
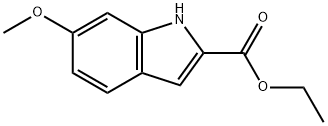
15050-04-1
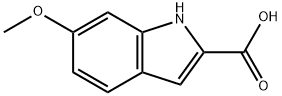
16732-73-3
Ethyl 6-methoxy-1H-indole-2-carboxylate (5.0 g) was dissolved in tetrahydrofuran (90 mL), lithium hydroxide (2.33 g) and water (30 mL) were added and the reaction was stirred for 16 hours at room temperature. After completion of the reaction, the reaction mixture was acidified with 10% hydrochloric acid solution, diluted with water and subsequently extracted with ethyl acetate. The organic phase was washed with brine, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give 6-methoxy-1H-indole-2-carboxylic acid (4.60 g). The resulting carboxylic acid (4.64 g) was dissolved with 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (5.60 g) in dichloromethane (200 mL), N-methylpiperazine (3.23 mL) was added, and the reaction was stirred for 16 hours at room temperature. The reaction mixture was diluted with dichloromethane (200 mL), washed sequentially with water, saturated sodium bicarbonate solution and brine, the organic phase was dried with anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel column chromatography (eluent: 0-10% 2M ammonia in methanol solution/dichloromethane) to give (6-methoxy-1H-indol-2-yl)-(4-methylpiperazin-1-yl)-methanone (6.60 g). The product (0.16 g) was dissolved in dichloromethane (10 mL) and 1M boron tribromide solution (1.5 mL) was added slowly and dropwise at room temperature, followed by heating the reaction mixture to reflux overnight. After cooling, the reaction was quenched with saturated sodium bicarbonate solution and extracted with dichloromethane. The organic phase was washed with brine, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The residue was purified by silica gel column chromatography (eluent: 0-10% 2M ammonia in methanol solution/dichloromethane) to give 6-methoxyindole-2-carboxylic acid as final product.
References
[1] Patent: US2003/207893, 2003, A1
[2] Patent: WO2004/22061, 2004, A1. Location in patent: Page/Page column 71
[3] Patent: EP1373204, 2016, B1. Location in patent: Paragraph 0216
[4] Bioorganic and Medicinal Chemistry Letters, 2017, vol. 27, # 9, p. 1943 - 1948
[5] Patent: CN106478606, 2017, A. Location in patent: Paragraph 0122; 0123; 0128; 0129
6-METHOXY-1H-INDOLE-2-CARBOXYLIC ACID Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| career henan chemical co | +86-0371-86658258 +8613203830695 | sales@coreychem.com | China | 29819 | 58 |
| Alchem Pharmtech,Inc. | 8485655694 | sales@alchempharmtech.com | United States | 63687 | 58 |
| Wuhan Chemwish Technology Co., Ltd | 027-67849912 | sales@chemwish.com | CHINA | 10821 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-29-81139210 +86-18192627656 | 1059@dideu.com | China | 3590 | 58 |
| Neostar United (Changzhou) Industrial Co., Ltd. | +86-0519-85551759 +8613506123987 | marketing1@neostarunited.com | China | 8828 | 58 |
| SIMAGCHEM CORP | +86-5922680277 +86-13806087780 | sale@simagchem.com | China | 17346 | 58 |
| Hubei Ipure Biology Co., Ltd | +8613367258412 | ada@ipurechemical.com | China | 10236 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418671 +8618949823763 | sales@tnjchem.com | China | 34563 | 58 |
| HONG KONG IPURE BIOLOGY CO.,LIMITED | 86 18062405514 18062405514 | ada@ipurechemical.com | CHINA | 3461 | 58 |
| ANHUI WITOP BIOTECH CO., LTD | +8615255079626 | eric@witopchemical.com | China | 23541 | 58 |
View Lastest Price from 6-METHOXY-1H-INDOLE-2-CARBOXYLIC ACID manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2025-12-12 | 6-Methoxyindole-2-carboxylic acid
16732-73-3
|
US $0.01-1.00 / KG | 1KG | 99% | 20 tons | Shaanxi Dideu Medichem Co. Ltd | |
 |
2019-07-06 | 6-METHOXY-1H-INDOLE-2-CARBOXYLIC ACID
16732-73-3
|
US $1.00 / kg | 1kg | 95%-99% | as request | Career Henan Chemical Co |
-

- 6-Methoxyindole-2-carboxylic acid
16732-73-3
- US $0.01-1.00 / KG
- 99%
- Shaanxi Dideu Medichem Co. Ltd
-

- 6-METHOXY-1H-INDOLE-2-CARBOXYLIC ACID
16732-73-3
- US $1.00 / kg
- 95%-99%
- Career Henan Chemical Co
16732-73-3(6-METHOXY-1H-INDOLE-2-CARBOXYLIC ACID)Related Search:
1of4





