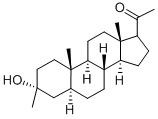
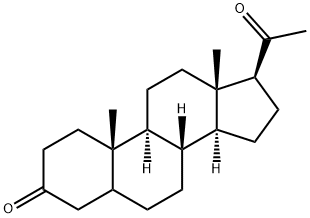
GANAXOLONE
- CAS No.
- 38398-32-2
- Chemical Name:
- GANAXOLONE
- Synonyms
- GANAXOLONE;Fampridine Impurity 80;5α-PREGNAN-3β-METHYL-3α-;Noradrenaline Impurity 69;5α-PREGNAN-3β-METHYL-3α-OL-20-ONE;3α-hydroxy-3β-methyl-5α-pregnan-20-one;(3α,5α)-3-Hydroxy-3-methylpregnan-20-one;(3α,5α)-3-Hydroxy-3-methyl-pregnan-20-one;Pregnan-20-one, 3-hydroxy-3-methyl-, (3α,5α)-;1-((3R,5S,8R,9S,10S,13S,14S,17S)-3-Hydroxy-3,10,13-trimethylhexadecahydro-1H-cyclopenta[a]phenanthren-17-yl)ethanone
- CBNumber:
- CB8498146
- Molecular Formula:
- C22H36O2
- Molecular Weight:
- 332.52
- MDL Number:
- MFCD09971088
- MOL File:
- 38398-32-2.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| Ganaxolone solid | G7795 | 5mg | $217 |
| Ganaxolone solid | G7795 | 25mg | $746 |
| Ganaxolone ≥98% | 20919 | 1mg | $36 |
| Ganaxolone ≥98% | 20919 | 5mg | $120 |
| Ganaxolone ≥98% | 20919 | 10mg | $169 |
| Melting point | 190-192° |
|---|---|
| alpha | D +103° |
| Boiling point | 434.8±18.0 °C(Predicted) |
| Density | 1.036±0.06 g/cm3(Predicted) |
| storage temp. | 2-8°C |
| solubility | DMSO: ~4.2 mg/mL |
| pka | 15.18±0.70(Predicted) |
| form | solid |
| color | white |
| InChI | InChI=1/C22H36O2/c1-14(23)17-7-8-18-16-6-5-15-13-20(2,24)11-12-21(15,3)19(16)9-10-22(17,18)4/h15-19,24H,5-13H2,1-4H3/t15-,16-,17,18-,19-,20+,21-,22+/s3 |
| InChIKey | PGTVWKLGGCQMBR-TVQBYSQTNA-N |
| SMILES | [C@@]12([H])CC[C@@]3([H])C[C@@](O)(C)CC[C@]3(C)[C@@]1([H])CC[C@]1(C)C(CC[C@@]21[H])C(=O)C |&1:0,4,7,12,14,18,23,r| |
| FDA UNII | 98WI44OHIQ |
| UNSPSC Code | 12352200 |
| NACRES | NA.77 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|---|---|
| Signal word | Warning |
| Hazard statements | H302+H312+H332 |
| Safety Statements | 22-24/25 |
| WGK Germany | 3 |
| RTECS | TU4384700 |
| Storage Class | 11 - Combustible Solids |
GANAXOLONE price More Price(19)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | G7795 | Ganaxolone solid | 38398-32-2 | 5mg | $217 | 2026-04-30 | Buy |
| Sigma-Aldrich | G7795 | Ganaxolone solid | 38398-32-2 | 25mg | $746 | 2026-04-30 | Buy |
| Cayman Chemical | 20919 | Ganaxolone ≥98% | 38398-32-2 | 1mg | $36 | 2026-04-30 | Buy |
| Cayman Chemical | 20919 | Ganaxolone ≥98% | 38398-32-2 | 5mg | $120 | 2026-04-30 | Buy |
| Cayman Chemical | 20919 | Ganaxolone ≥98% | 38398-32-2 | 10mg | $169 | 2026-04-30 | Buy |
GANAXOLONE Chemical Properties,Uses,Production
Description
Ganaxolone is a 3β-methylated synthetic analog of the neurosteroid allopregnanolone that allosterically enhances GABAA receptor current. At nanomolar concentrations, it can potentiate GABA-evoked currents at GABAA receptor complexes containing α1, α2, α3, β2, and γ2L subunits expressed in Xenopus oocytes. Ganaxolone has been shown to have a broad range of antiseizure activity in animal epilepsy models and human clinical trials.
Uses
Treatment of epilepsy and migraine.
Uses
Ganaxolone, is synthetic neuroactive steroid, which possess sedative, anxiolytic, and anticonvulsant properties. It is also a potent and selective positive allosteric modulator of GABAA receptors. It is also shown to be used for the treatment of epilepsy.
Definition
ChEBI: Ganaxolone is a corticosteroid hormone.
Application
Ganaxolone (marketed as Ztalmy) is a first-in-class neuroactive steroid that acts as a positive modulator of gamma-aminobutyric acid (GABA) A receptors for the treatment of seizures associated with cyclooxygenase-dependent kinase-like 5 (CDKL5) deficiency (CDD).
Biological Activity
Potent positive allosteric modulator of GABA A receptors. Enhances GABA-evoked chloride currents in Xenopus oocytes expressing GABA A receptors (EC 50 values are 94, 122 and 213 nM for α 2 β 1 γ 2 L , α 3 β 1 γ 2 L and α 1 β 1 γ 2 L receptors respectively). Exerts anticonvulsive effects in a broad range of animal seizure models.
Biochem/physiol Actions
Ganaxolone (3alpha-hydroxy-3beta-methyl-5alpha-pregnane-20-one) is a positive allosteric modulator of the GABAA receptor subtype; synthetic analog of the endogenous neurosteroid allopregnanolone; effective against chemically induced seizures in rats and mice. Ganaxolone is an orally active analog of allopregnanolone that is not converted to the hormonally active 3-keto form. The enhanced anticonvulsant potency of ganaxolone after neurosteroid withdrawal supports the use of ganaxolone as a specific treatment for perimenstrual catamenial epilepsy.
Mechanism of action
Ganaxolone works by positively modulating GABAA receptors, the major inhibitory neurotransmitter receptors in the central nervous system. This modulatory effect helps reduce the frequency and severity of seizures associated with CDD.
Synthesis
The synthesis begins with protection of the keto functionality in the natural steroid pregnenolone 10.1. Using acid catalysis, reaction with boiling ethylene glycol in toluene forms the acetal 10.2 in 88% yield. Oxidation of 10.2 to form the ketone 10.3 has been accomplished by a variety of methods, including Swern, Dess-Martin, and TPAP conditions. The oxidation process depicted in Figure 3.1 demonstrates the formation of the ketone 10.3 using calcium hypochlorite and TEMPO. Next, methylation is accomplished by the addition of MeMgBr in the presence of lithium chloride and ferric chloride to afford the tertiary alcohol 10.4. Finally, removal of the acetal protecting group by treatment with iodine in dichloromethane and acetone affords ganaxolone 10 in 98% yield over the last two steps.
storage
+4°C
References
[1] R B CARTER. Characterization of the anticonvulsant properties of ganaxolone (CCD 1042; 3alpha-hydroxy-3beta-methyl-5alpha-pregnan-20-one), a selective, high-affinity, steroid modulator of the gamma-aminobutyric acid(A) receptor.[J]. Journal of Pharmacology and Experimental Therapeutics, 1997, 280 3: 1284-1295.
GANAXOLONE Preparation Products And Raw materials
GANAXOLONE Suppliers
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Hunan Yuanye Pharmaceutical Co., Ltd. | +8615857615745 | 2675013845@qq.com | China | 87 | 58 |
| Hangzhou Hyper Chemicals Limited | +86-0086-57187702781 +8613675893055 | info@hyper-chem.com | China | 310 | 58 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | factory@coreychem.com | China | 29794 | 58 |
| Zhejiang J&C Biological Technology Co.,Limited | +1-2135480471 +1-2135480471; | sales@sarms4muscle.com | China | 10473 | 58 |
| InvivoChem | +1-708-310-1919 +1-13798911105 | sales@invivochem.cn | United States | 6391 | 58 |
| Hangzhou MolCore BioPharmatech Co.,Ltd. | +86-057181025280; +8617767106207 | sales@molcore.com | China | 49734 | 58 |
| TargetMol Chemicals Inc. | +1-781-999-5354; | support@targetmol.com | United States | 39040 | 58 |
| Zibo Hangyu Biotechnology Development Co., Ltd | +86-0533-2185556 +8615965530500 | nickzhang@hangyubiotech.com | China | 8612 | 58 |
| Shaanxi Dideu New Materials Co. Ltd | +86-18192503167 18192503167; | 1036@dideu.com | China | 7917 | 58 |
| Moxin Chemicals | +86-17320513646 +8617320513646 | anna@molcoo.com | China | 10000 | 58 |
View Lastest Price from GANAXOLONE manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2026-04-24 | Ganaxolone
38398-32-2
|
US $0.00-0.00 / Gram | 1Gram | 98% | 50KG | Hangzhou Hyper Chemicals Limited | |
 |
2025-12-01 | GANAXOLONE
38398-32-2
|
US $0.00 / kg | 1kg | 99% | 10000KGS | Shaanxi Dideu New Materials Co. Ltd | |
 |
2020-01-10 | GANAXOLONE
38398-32-2
|
US $0.01 / KG | 1KG | 98%; 99% | 500g;1kg; 25kg | Career Henan Chemical Co |
-

- Ganaxolone
38398-32-2
- US $0.00-0.00 / Gram
- 98%
- Hangzhou Hyper Chemicals Limited
-

- GANAXOLONE
38398-32-2
- US $0.00 / kg
- 99%
- Shaanxi Dideu New Materials Co. Ltd
-

- GANAXOLONE
38398-32-2
- US $0.01 / KG
- 98%; 99%
- Career Henan Chemical Co