| 구조 |
화학 이름 |
CAS |
MF |
 |
Gastric soluble film coating premix (medicinal excipient) |
|
|
 |
Diethyl phthalate (medicinal excipients) |
|
|
 |
Film coating premix (enteric soluble) (medicinal excipients) |
|
|
 |
Film coating agent (stomach-soluble type) (medicinal excipients) |
|
|
 |
Aqueous dispersion of methacrylic acid-ethyl acrylate copolymer (pharmaceutical excipient) |
|
|
 |
Pharmaceutical grade titanium dioxide, food grade titanium dioxide (titanium dioxide) |
|
|
 |
Silica (SYLYSIA 350) (medicinal excipients) |
|
|
 |
Soy lecithin (medicinal excipients for injection) |
|
|
 |
Tyloxapol (medicinal excipients) |
|
|
 |
Sodium lauryl sulfate (medicinal excipients) |
|
|
 |
Carboxymethyl starch sodium |
|
|
 |
White beeswax (medicinal excipients) |
|
|
 |
Soybean oil (medicinal excipients) |
|
|
 |
TX-10 |
|
|
 |
Capo 940 |
|
|
 |
엔도-노르보르네올 |
497-36-9 |
C7H12O |
 |
Aspartame (medicinal excipients) |
|
|
 |
Ethanol (75%) (medicinal excipients) |
|
C2H6O |
 |
Pharmaceutical grade polyethylene glycol 400 |
|
|
 |
Carboxymethylcellulose sodium (medicinal excipients) |
|
|
 |
Mixed fatty acid glyceride (hard fat) (medicinal excipients) |
|
|
 |
2, 5-Tetrahydrofurandimethanol Fatty Acid Diesters |
|
|
 |
Dextrin pharmaceutical grade |
|
|
 |
Hollow capsule |
|
|
 |
Pharmaceutical excipient magnesium stearate |
|
|
 |
Medicinal blank pill |
|
|
 |
Microcrystalline cellulose pellets (medicinal excipients) |
|
|
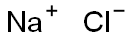 |
Sodium chloride (for injection) (medicinal excipients) |
|
ClNa |
 |
Sucrose Octasulfate Octatriethylamine Salt (>90%) |
869561-07-9 |
C18H37NO35S8 |
 |
Povidone K 30 |
|
|
 |
Pharmaceutical grade urea |
|
|
 |
Corn oil (pharmaceutical excipient) |
|
|
 |
medicine adhesive |
|
|
 |
iron oxide,purple for pharmaceutical |
|
|
 |
Medicinal tannin |
|
|
 |
Medicinal bleaching activated carbon |
|
|
 |
Polyacrylic acid resin III |
|
|
 |
Silica (SYLYSIA 350) (medicinal excipients) |
|
|
 |
Carmellose (medicinal excipients) |
|
|
 |
Potassium sorbate (medicinal excipients) |
|
|
 |
Carbon dioxide (medicinal excipients) |
|
|
 |
Benzoic acid (medicinal excipients) |
|
|
 |
Recombinant human albumin (pharmaceutical excipient) |
|
|
 |
Silica (medicinal excipients) |
|
|
 |
Di-erucylphosphatidylcholine (medicinal excipient) |
|
|
 |
Adenine (medicinal excipients) |
|
|
 |
Amorphous sorbitol solution (medicinal excipients) |
|
|
 |
Brown iron oxide (medicinal excipients) |
|
|
 |
Peony phosphatidylethanolamine (for injection) (medicinal excipients) |
|
|
 |
Histidine (medicinal excipients) |
|
|
 |
Medicinal ethanol |
|
|
 |
Medicinal alcohol |
|
|
 |
Stevioside (pharmaceutical excipient) |
|
|
 |
Starch lactose (medicinal excipients) |
|
|
 |
Medicinal ammonium chloride |
|
|
 |
Capsule with gelatin |
|
|
 |
마이크로결정LINE 왁스 |
63231-60-7 |
unspecified |
 |
Pharmaceutical film coated premixed excipients |
|
|
 |
Rhubarb (medicinal rhubarb) |
|
|
 |
Carbomer homopolymer type A 971P NF (medicinal excipients) |
|
|
 |
Hollowcapsule |
|
|
 |
Hypromellose phthalate (medicinal excipients) |
|
|
 |
Medicinal carbon black |
|
|
 |
MANDRAGORA OFFICINARUM |
|
|
 |
Pharmaceutical grade water-soluble starch |
|
|
 |
하이드록시프로필 메틸셀룰로스 |
9004-65-3 |
C3H7O* |
 |
Medicinal sodium bicarbonate |
|
|
 |
Microcrystalline cellulose (medicinal excipients) |
|
|
 |
Thymol (medicinal excipients) |
|
|
 |
Calcium chloride (medicinal excipients) |
|
|
 |
Hydroxyethyl ester (medicinal excipients) |
|
|
 |
Sucrose stearate (medicinal excipients) |
|
|
 |
Glycolide lactide copolymer (5050) (for injection) (medicinal excipients) |
|
|
 |
Anhydrous calcium hydrogen phosphate (medicinal excipients) |
|
|
 |
Benzalkonium chloride (medicinal excipients) |
|
|
 |
Microcrystalline cellulose M102CG (medicinal excipients) |
|
|
 |
Ascorbyl palmitate (medicinal excipient) |
|
|
 |
Potassium hydroxide (pharmaceutical excipient) |
|
|
 |
PSZ104 |
|
|
 |
펠리토리추출물 |
84012-32-8 |
|
 |
Capsule release agent |
|
|
 |
Maltose (pharmaceutical excipient) |
|
|
 |
Pharmaceutical grade corn starch |
|
|
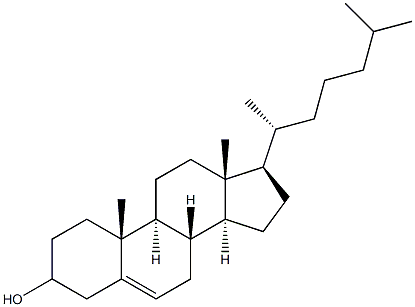 |
Cholesterol (for injection) (medicinal excipients) |
|
C27H46O |
 |
Lactose (medicinal excipients) |
|
|
 |
Triethanolamine (medicinal excipients) |
|
|
 |
Disodium edetate (medicinal excipients) |
|
|
 |
Sodium benzoate (medicinal excipients) |
|
|
 |
Silica (SYLYSIA 350FCP) (medicinal excipients) |
|
|
 |
Methyl cellulose (medicinal excipients) |
|
|
 |
Soybean phospholipid (medicinal excipient) |
|
|
 |
Croscarmellose sodium (medicinal excipients) |
|
|
 |
Lactose (granular type) (medicinal excipients) |
|
C12H22O11 |
 |
Colloidal silica (medicinal excipients) |
|
|
 |
Methanol (medicinal excipients) |
|
|
 |
Disodium edetate (for injection) (medicinal excipients) |
|
C10H14N2Na2O8 |
 |
Ethyl acetate (medicinal excipients) |
|
|
 |
Emulsifier 400 do |
|
|
 |
Hydroxyethyl Cellulose |
|
|
 |
Microcrystalline cellulose M102CG (medicinal excipients) |
|
|