IBUPROFEN SODIUM SALT manufacturers
- Ibuprofen sodium
-

- $0.00 / 25mg
-
2026-04-20
- CAS:31121-93-4
- Min. Order:
- Purity:
- Supply Ability: 10g
- IBUPROFEN SODIUM SALT
-

- $0.00 / 1kg
-
2025-08-22
- CAS:31121-93-4
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 500kg
|
| | IBUPROFEN SODIUM SALT Basic information |
| Product Name: | IBUPROFEN SODIUM SALT | | Synonyms: | IBUPROFEN SODIUM SALT;alpha-methyl-4-(2-methylpropyl)-benzeneaceticacisodiumsalt;ibuprofensodium;p-isobutylhydratropicacidsodiumsalt;ALPHA-METHYL-4-[2-METHYLPROPYL]BENZENEACETIC ACID SODIUM SALT;ALPHA-METHYL-4-(ISOBUTYL)PHENYLACETIC ACID SODIUM SALT;p-isobutyl-hydratropicacisodiumsalt;sodium 2-(4-isobutylphenyl)propionate | | CAS: | 31121-93-4 | | MF: | C13H17NaO2 | | MW: | 228.26 | | EINECS: | 250-477-6 | | Product Categories: | | | Mol File: | 31121-93-4.mol | 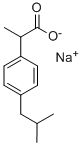 |
| | IBUPROFEN SODIUM SALT Chemical Properties |
| Melting point | 110-112 °C | | storage temp. | Hygroscopic, -20°C Freezer, Under inert atmosphere | | solubility | H2O: 100 mg/mL, may be clear to slightly hazy | | form | Solid | | color | White to Off-White | | Water Solubility | H2O: 100mg/mL, clear to slightly hazy | | Stability: | Stable. Incompatible with strong oxidizing agents | | Major Application | forensics and toxicology
pharmaceutical (small molecule) | | InChI | 1S/C13H18O2.Na/c1-9(2)8-11-4-6-12(7-5-11)10(3)13(14)15;/h4-7,9-10H,8H2,1-3H3,(H,14,15);/q;+1/p-1 | | InChIKey | PTTPUWGBPLLBKW-UHFFFAOYSA-M | | SMILES | [Na+].CC(C)Cc1ccc(cc1)C(C)C([O-])=O | | CAS DataBase Reference | 31121-93-4 |
| Safety Statements | 22-24/25 | | WGK Germany | 3 | | RTECS | MU6641000 | | Storage Class | 11 - Combustible Solids |
| | IBUPROFEN SODIUM SALT Usage And Synthesis |
| Chemical Properties | white powder | | Uses | Ibuprofen ((±)-Ibuprofen) sodium is an orally active, selective COX-1 inhibitor with an IC50 value of 13 μM. Ibuprofen sodium inhibits cell proliferation, angiogenesis, and induces cell apoptosis. Ibuprofen sodium is a nonsteroidal anti-inflammatory agent and a nitric oxide (NO) donor. Ibuprofen sodium can be used in the research of pain, swelling, inflammation, infection, immunology, cancers[1][2][5][8]. | | Application | Ibuprofen sodium salt is a useful research chemical. | | Biological Activity | Cyclooxygenase (COX) inhibitor th at has greater activity against COX-1 than against COX-2. | | in vivo | Ibuprofen sodium (fed in animal feedings, 300 mg/kg, 14 days) reduces overall tumor growth and enhances anti-tumor immune characteristics without adverse autoimmune reactions in a model of postpartum breast cancer[5].
Ibuprofen sodium (subcutaneous injection, 60 mg/kg, every second day for 15 days) reduces the risk of neuropathy in a rat model of chronic Oxaliplatininduced peripheral neuropathy[6].
Ibuprofen sodium (oral administration, 20 mg/kg, every 12 hours, 5 doses total) decreases muscle growth (average muscle fiber cross-sectional area) without affecting regulation of supraspinatus tendon adaptions to exercise[7].
Ibuprofen sodium (oral administration, 35 mg/kg, twice daily) attenuates the Inflammatory response to pseudomonas aeruginosa in a rat model of chronic pulmonary infection[8].
| Animal Model: | Syngeneic (D2A1) orthotopic Balb/c mouse model of PPBC (postpartum)[5] | | Dosage: | 300 mg/kg, daily for 14 days | | Administration: | Fed in animal feedings (added to pulverized standard chow and mixed dry, then mixed with water, made into chow pellets and dried thoroughly) | | Result: | Suppresed tumor growth, reduced presence of immature monocytes and increased numbers of T cells.
Enhanced Th1 associated cytokines as well as promoted tumor border accumulation of T cells.
|
| Animal Model: | Oxaliplatininduced peripheral neuropathy[6] | | Dosage: | 60 mg/kg, every second day for 15 days | | Administration: | Subcutaneous injection | | Result: | Lowered sensory nerve conduction velocity (SNCV). |
| | IC 50 | COX-1: 13 μM (IC50); COX-2: 370 μM (IC50) | | References | [1] Noreen Y, et al. Development of a radiochemical cyclooxygenase-1 and -2 in vitro assay for identification of natural products as inhibitors of prostaglandin biosynthesis. J Nat Prod. 1998 Jan;61(1):2-7. DOI:10.1021/np970343j
[2] Hassan Akrami, et al. Inhibitory effect of ibuprofen on tumor survival and angiogenesis in gastric cancer cell. Tumour Biol. 2015 May;36(5):3237-43. DOI:10.1007/s13277-014-2952-3
[3] Sharon M Rymut, et al. Ibuprofen regulation of microtubule dynamics in cystic fibrosis epithelial cells. Am J Physiol Lung Cell Mol Physiol. 2016 Aug 1;311(2):L317-27. DOI:10.1152/ajplung.00126.2016
[4] Emmanuelle Bignon, et al. Ibuprofen and ketoprofen potentiate UVA-induced cell death by a photosensitization process. Sci Rep. 2017 Aug 21;7(1):8885. DOI:10.1038/s41598-017-09406-8
[5] Nathan D Pennock, et al. Ibuprofen supports macrophage differentiation, T cell recruitment, and tumor suppression in a model of postpartum breast cancer. J Immunother Cancer. 2018 Oct 1;6(1):98. DOI:10.1186/s40425-018-0406-y
[6] Thomas Krigrd, et al. Protective effect of ibuprofen in a rat model of chronic oxaliplatin-induced peripheral neuropathy. Exp Brain Res. 2019 Oct;237(10):2645-2651. DOI:10.1007/s00221-019-05615-x
[7] Sarah Ilkhanipour Rooney, et al. Ibuprofen Differentially Affects Supraspinatus Muscle and Tendon Adaptations to Exercise in a Rat Model. Am J Sports Med. 2016 Sep;44(9):2237-45. DOI:10.1177/0363546516646377
[8] M W Konstan, et al. Ibuprofen attenuates the inflammatory response to Pseudomonas aeruginosa in a rat model of chronic pulmonary infection. Implications for antiinflammatory therapy in cystic fibrosis. Am Rev Respir Dis. 1990 Jan;141(1):186-92. DOI:10.1164/ajrccm/141.1.186 |
| | IBUPROFEN SODIUM SALT Preparation Products And Raw materials |
|