- Volitinib
-

- $0.00 / 1kg
-
2026-04-23
- CAS:1313725-88-0
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 20tons
- Savolitinib
-

- $42.00 / 1mg
-
2026-04-22
- CAS:1313725-88-0
- Min. Order:
- Purity: 98.12%
- Supply Ability: 10g
- Volitinib USP/EP/BP
-

- $1.10 / 1g
-
2025-11-18
- CAS:1313725-88-0
- Min. Order: 1g
- Purity: 99.9%
- Supply Ability: 100 Tons Min
|
| | Savolitinib Basic information |
| Product Name: | Savolitinib | | Synonyms: | AZD6094,Volitinib, HMPL-504, Savolitinib;1-[(1S)-1-Imidazo[1,2-a]pyridin-6-ylethyl]-6-(1-methyl-1H-pyrazol-4-yl)-1H-1,2,3-triazolo[4,5-b]pyrazine;(S)-1-(1-(imidazo[1,2-a]pyridin-6-yl)ethyl)-6-(1-methyl-1H-pyrazol-4-yl)-1H-[1,2,3]triazolo[4,5-b]pyrazine;Volitinib;Savolitinib;HMPL-504(AZD6094, Volitinib);EOS-60799;AZD-6094) | | CAS: | 1313725-88-0 | | MF: | C17H15N9 | | MW: | 345.36 | | EINECS: | | | Product Categories: | API | | Mol File: | 1313725-88-0.mol | 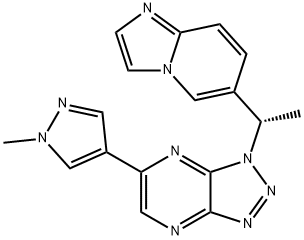 |
| | Savolitinib Chemical Properties |
| Melting point | 205-207°C | | density | 1.55±0.1 g/cm3(Predicted) | | storage temp. | -20°C Freezer | | solubility | DMSO (Slightly), Methanol (Slightly) | | form | Solid | | pka | 5.67±0.50(Predicted) | | color | Pale Beige | | InChI | InChI=1S/C17H15N9/c1-11(12-3-4-15-18-5-6-25(15)10-12)26-17-16(22-23-26)19-8-14(21-17)13-7-20-24(2)9-13/h3-11H,1-2H3/t11-/m0/s1 | | InChIKey | XYDNMOZJKOGZLS-NSHDSACASA-N | | SMILES | C12N=NN([C@H](C3=CN4C=CN=C4C=C3)C)C1=NC(C1=CN(C)N=C1)=CN=2 | | CAS DataBase Reference | 1313725-88-0 |
| | Savolitinib Usage And Synthesis |
| Description | Savolitinib selectively inhibits the MET receptor, blocking the PI3K/AKT/MAPK-signaling
pathway as well as downregulating MYC. | | Uses | Savolitinib is a potent and highly selective c-Met inhibitor. Savoltinib demonstrated anti-tumor efficacy in a panel of cMet-dysregulated gastric cancer PDX models. Savolitinib also demonstrated good antitmuor activities. | | Clinical Use | It is currently being evaluated
in phase I clinical trials in combination with EGFR TKIs in NSCLC patients. | | Synthesis | The synthesis of savolitinib started with 6-amino-3-carboxypyridine (20.1), which was treated with chloroacetaldehyde to generate the imidazopyridine 20.2. The carboxylic acid was converted to the Weinreb amide 20.3 and treated with methylmagnesium bromide to give the methyl ketone 20.4. The ketone was converted to the chiral amine 20.5 using a commercially available aminotransferase with the cofactors pyridoxal phosphate and isopropylamine as a nitrogen source. After process workup, the amine was isolated as the dihydrochloride salt (20.5). The free base was obtained by treatment with alkaline conditions, which was subsequently reacted with commercially available 3,5-dibromopyrazin-2-amine (20.6) to give the secondary amine 20.7. Next, nitrosyl ion formation and cyclization gave the 1,2,3-triazole 20.8. Finally, Suzuki coupling with 20.9 completed the construction of savolitinib (20). The boronic acid ester 20.9 was obtained from 4-bromo-1-methyl-1H-pyrazole by lithium-halogen exchange and workup with triisopropylborate quenching.
 | | in vivo | Savolitinib (Compound 28; 1-10.0 mg/kg; oral administration; daily; for 21 days; athymic nude mice) demonstrates dose-dependent tumor growth inhibition in a U87MG subcutaneous xenograft model. In addition, none of the mice in the dosing groups exhibits body weight loss during the experiment[1]. | Animal Model: | U87MG xenograft model in athymic nude mice[1] | | Dosage: | 1 mg/kg, 2.5 mg/kg and 10.0 mg/kg | | Administration: | Oral administration; daily; for 21 days | | Result: | Demonstrated dose-dependent tumor growth inhibition in a U87MG subcutaneous xenograft model.
|
|
| | Savolitinib Preparation Products And Raw materials |
|