Beclabuvir manufacturers
- Beclabuvir
-

- $137.00 / 1mg
-
2026-04-20
- CAS:958002-33-0
- Min. Order:
- Purity: 99.93%
- Supply Ability: 10g
- Beclabuvir
-

- $137.00 / 1mg
-
2026-04-20
- CAS:958002-33-0
- Min. Order:
- Purity: 99.93%
- Supply Ability: 10g
|
| | Beclabuvir Basic information |
| Product Name: | Beclabuvir | | Synonyms: | Beclabuvir;Beclabuvir(BMS-791325);(1aR,12bS)-8-cyclohexyl-N-[(dimethylamino)sulfonyl]-1,1a,2,12b-tetrahydro-11-methoxy-1a-[(3-methyl-3,8-diazabicyclo[3.2.1]oct-8-yl)carbonyl]-cycloprop[d]indolo[2,1-a][2]benzazepine-5-carboxamide;Cycloprop[d]indolo[2,1-a][2]benzazepine-9-carboxamide, 12-cyclohexyl-N-[(dimethylamino)sulfonyl]-4b,5,5a,6-tetrahydro-3-methoxy-5a-[(3-methyl-3,8-diazabicyclo[3.2.1]oct-8-yl)carbonyl]-, (4bS,5aR)- | | CAS: | 958002-33-0 | | MF: | C36H45N5O5S | | MW: | 659.84 | | EINECS: | | | Product Categories: | | | Mol File: | 958002-33-0.mol | 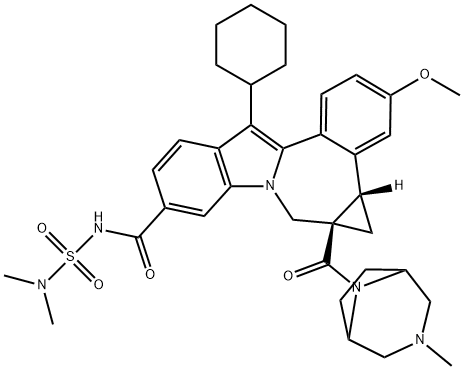 |
| | Beclabuvir Chemical Properties |
| density | 1.45±0.1 g/cm3(Predicted) | | storage temp. | Store at -20°C | | solubility | DMSO:30.0(Max Conc. mg/mL);45.46(Max Conc. mM) | | form | Solid | | pka | 4.44±0.40(Predicted) | | color | White to off-white |
| | Beclabuvir Usage And Synthesis |
| Description | Beclabuvir is a non-nucleoside,
nonstructural protein 5B (NS5B) polymerase inhibitor
approved in Japan as part of a fixed-dose combination product
for the treatment of hepatitis C virus (HCV). Upon
administration and after intracellular uptake, the drug binds
to the allosteric, noncatalytic “Thumb 1” site of NS5B resulting
in a decreased rate of viral RNA synthesis and replication.4
Beclabuvir is combined with asunaprevir and declatasvir (both
approved in 2014) and was discovered and developed by
Bristol-Myers Squibb. | | Uses | Beclabuvir is an allosteric inhibitor that binds to thumb site 1 of the hepatitis C virus (HCV) NS5B RNA-dependent RNA polymerase, and inhibits recombinant NS5B proteins from HCV genotypes 1, 3, 4, and 5 with IC50 of < 28 nM[1][2]. | | Synthesis | The syntheses of asunaprevir and declatasvir were described
in an earlier review article.Condensation of
indole-6-carboxylic acid (1) with cyclohexanone under basic
conditions gave acid 2 in quantitative yield. Hydrogenation of
the double bond in 2 using Pearlmanˉs catalyst was followed by
esterification to give ester 3 in high yield. Bromination of the
indole at the 2-position was accomplished with pyridinium
tribromide, and this was followed by saponification to provide
acid 4. Treatment of 4 with carbonyldiimidazole (CDI)
followed by N,N- dimethylsulfamide and 1, 8 -
diazabicyclo[5.4.0]undec-7-ene (DBU) gave compound 5 in
74% yield. Suzuki coupling of 5 with commercial boronic acid 6
provided intermediate 7, which converted to hemiaminal 8
upon continued heating in 61% yield. Compound 8 was then
treated with methyl 2-(dimethoxyphosphoryl)acrylate (9) to
affect a tandem conjugate addition and HornerWadsworth
Emmons (HWE) olefination to give ester 10. Alternatively, the
Suzuki coupling reaction of 5 with 6 could be stopped at
intermediate 7, which could be treated with 9 to promote the tandem conjugate addition/HWE to give 10. Corey-Chaykovsky cyclopropanation of 10 using sodium hydride
and trimethylsulfoxonium iodide followed by chiral separation
provided cyclopropane 11 in good yield and >99% enantiomeric
excess (ee). Saponification of the methyl ester of 11
followed by coupling with 3-methyl-3,8-diazabicyclo[3.2.1]-
octane dihydrochloride (12) gave beclabuvir (I) in high yield.
 | | in vivo | The combination of beclabuvir with asunaprevir and daclatasvir achieves very high rates of viral eradication (about 90%) in patients infected with HCV genotype 1, which is the most common genotype worldwide[1]. | | References | [1] Gentile I, et al. Beclabuvir for the treatment of hepatitis C. Expert Opin Investig Drugs. 2015;24(8):1111-21 DOI:10.1517/13543784.2015.1059820
[2] Tatum H, et al. A randomized, placebo-controlled study of the NS5B inhibitor beclabuvir with peginterferon/ribavirin for HCV genotype 1. J Viral Hepat. 2015 Aug;22(8):658-64. DOI:10.1111/jvh.12372 |
| | Beclabuvir Preparation Products And Raw materials |
|