- Fevipiprant
-
-
2026-05-13
- CAS:872365-14-5
- Min. Order:
- Purity: 0.99
- Supply Ability:
- Fevipiprant
-

- $29.00 / 5mg
-
2026-05-11
- CAS:872365-14-5
- Min. Order:
- Purity: 99.32%
- Supply Ability: 10g
- Fevipiprant
-
- $2300.00 / 10gram
-
2025-08-22
- CAS:872365-14-5
- Min. Order: 10gram
- Purity: 99%
- Supply Ability: 20 tons
|
| | Fevipiprant Basic information |
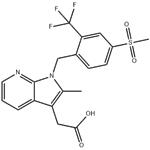
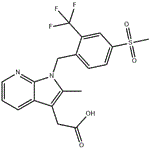
| Product Name: | Fevipiprant | | Synonyms: | QAW039;Fevipiprant;Fevipipran;NVP-QAW039;Fevipiprant(NVP-QAW039);Fevipiprant (QAW039);2-Methyl-1-[[4-(methylsulfonyl)-2-(trifluoromethyl)phenyl]methyl]-1H-pyrrolo[2,3-b]pyridine-3-acetic acid;NVP-QAW039; NVP-QAW 039; NVP-QAW-039 QAW-039; QAW039; QAW 039; FEVIPIPRANT | | CAS: | 872365-14-5 | | MF: | C19H17F3N2O4S | | MW: | 426.41 | | EINECS: | | | Product Categories: | | | Mol File: | 872365-14-5.mol |  |
| | Fevipiprant Chemical Properties |
| Boiling point | 637.6±55.0 °C(Predicted) | | density | 1.44±0.1 g/cm3(Predicted) | | storage temp. | Store at -20°C | | solubility | DMF: 10 mg/mL; DMF:PBS(pH7.2) (1:2): 0.33 mg/mL; DMSO: 10 mg/mL; Ethanol: 1 mg/mL | | form | A crystalline solid | | pka | 3.33±0.30(Predicted) | | color | White to off-white | | InChI | InChI=1S/C19H17F3N2O4S/c1-11-15(9-17(25)26)14-4-3-7-23-18(14)24(11)10-12-5-6-13(29(2,27)28)8-16(12)19(20,21)22/h3-8H,9-10H2,1-2H3,(H,25,26) | | InChIKey | GFPPXZDRVCSVNR-UHFFFAOYSA-N | | SMILES | C12N(CC3=CC=C(S(C)(=O)=O)C=C3C(F)(F)F)C(C)=C(CC(O)=O)C1=CC=CN=2 |
| | Fevipiprant Usage And Synthesis |
| Uses | Fevipiprant is used in the oral treatment of asthma and has been successful in targeting severe eosinophilic asthma.Fevipiprant is a novel oral prostaglandin D2 receptor 2 (DP2; known as CRTh2) antagonist and currently in development for the treatment of severe asthma and atopic dermatitis. | | in vivo | Fevipiprant (10 mg/kg; in the drinking water) reduces CaCl2-induced AAA (abdominal aortic aneurysm) formation in mouse[3]. | Animal Model: | C57Bl/6 mice[3] | | Dosage: | 10 mg/kg | | Administration: | In the drinking water | | Result: | Efficiently reduced CaCl2-induced AAA formation with diminished elastin degradation, aortic macrophage infiltration, MPO accumulation and MCP-1 expression. |
| | IC 50 | DP2: 1.14 nM (Kd) |
| | Fevipiprant Preparation Products And Raw materials |
|