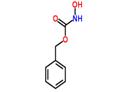
- Cbz-NH-OH
-
- $1.00 / 1g
-
2020-01-06
- CAS:3426-71-9
- Min. Order: 1g
- Purity: 98%
- Supply Ability: 100KG
|
| | BENZYL N-HYDROXYCARBAMATE Basic information |
| | BENZYL N-HYDROXYCARBAMATE Chemical Properties |
| Melting point | 65-70 °C(lit.) | | Boiling point | 295.73°C (rough estimate) | | density | 1.2917 (rough estimate) | | refractive index | 1.5570 (estimate) | | storage temp. | Keep in dark place,Sealed in dry,Room Temperature | | solubility | DMSO (Slightly), Methanol (Slightly) | | form | Solid | | pka | 9.20±0.23(Predicted) | | color | White | | Sensitive | Moisture Sensitive | | BRN | 1074034 | | InChI | InChI=1S/C8H9NO3/c10-8(9-11)12-6-7-4-2-1-3-5-7/h1-5,11H,6H2,(H,9,10) | | InChIKey | PQBSPTAPCMSZAA-UHFFFAOYSA-N | | SMILES | C(OCC1=CC=CC=C1)(=O)NO | | CAS DataBase Reference | 3426-71-9(CAS DataBase Reference) |
| Safety Statements | 22-24/25 | | WGK Germany | 3 | | RTECS | FB1600000 | | F | 21 | | HS Code | 29280000 | | Storage Class | 11 - Combustible Solids |
| | BENZYL N-HYDROXYCARBAMATE Usage And Synthesis |
| Chemical Properties | White Crystalline Solid | | Uses | N-(Benzyloxycarbonyl)hydroxylamine reacts with cyclohepta-1,3-diene to produce 6-oxa-7-aza-bicyclo[3.2.2]non-8-ene-7-carboxylic acid benzyl ester. | | Synthesis | The general procedure for the synthesis of N-(benzylcarbonyloxy)hydroxylamine from benzyl chloroformate was as follows: 92.9 g (0.876 mol) of sodium carbonate (Na2CO3) and 300 mL of pure water were added to a 1 L three-necked flask, and the sodium carbonate was completely dissolved by mechanical stirring. Subsequently, 46.83 g (0.674 mol) of hydroxylamine hydrochloride was added, and 220 mL of dichloromethane solution containing 100 g (0.587 mol) of benzyl chloroformate was stirred and slowly added dropwise at room temperature for about 4 hours. After the dropwise addition, stirring was continued at room temperature for 1 h. Then 300 mL of pure water was added to separate the organic and aqueous layers. The aqueous layer was extracted with dichloromethane (150 mL each time, 3 times in total), and all the dichloromethane phases were combined, washed with 300 mL of saturated saline, and then dried with 20.0 g of anhydrous sodium sulfate. The dried solution was distilled under reduced pressure at 45 °C to remove the solvent to give the crude product I. The crude product was dissolved in 100 mL of dichloromethane, ensuring complete dissolution, then cooled to 0 °C in an ice-water bath and 300 mL of petroleum ether was added slowly and dropwise. The solution was crystallized at 0 °C for 1 h. The crystalline product was washed with pre-cooled mixed solvent (200 mL, petroleum ether: dichloromethane = 3:1, v/v) and dried at 35 °C under reduced pressure. Finally, it was stirred at 0 °C for 5 h to give 63.0 g of white powdery solid in 64% overall yield. | | References | [1] Organic and Biomolecular Chemistry, 2009, vol. 7, # 21, p. 4531 - 4538
[2] Journal of Organic Chemistry, 2009, vol. 74, # 22, p. 8690 - 8694
[3] Journal of the American Chemical Society, 2010, vol. 132, # 37, p. 12862 - 12864
[4] Synthetic Communications, 2010, vol. 40, # 5, p. 642 - 646
[5] Helvetica Chimica Acta, 1991, vol. 74, # 8, p. 1653 - 1670 |
| | BENZYL N-HYDROXYCARBAMATE Preparation Products And Raw materials |
|