|
|
| | Firategrast Basic information |
| Product Name: | Firategrast | | Synonyms: | SB-683699;SB 683699;SB683699;CS-1588;SB-683699,Firategrast;Firategrast;[1,1'-Biphenyl]-4-propanoic acid, α-[(2,6-difluorobenzoyl)aMino]-4'-(ethoxyMethyl)-2',6'-diMethoxy-, (αS)-;SB 683699;SB683699;SB-683699 | | CAS: | 402567-16-2 | | MF: | C27H27F2NO6 | | MW: | 499.5 | | EINECS: | | | Product Categories: | | | Mol File: | 402567-16-2.mol | 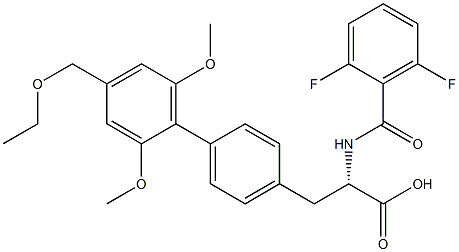 |
| | Firategrast Chemical Properties |
| Melting point | 154-155℃ (ethanol water ) | | Boiling point | 594.4±50.0 °C(Predicted) | | density | 1.261±0.06 g/cm3(Predicted) | | storage temp. | Store at -20°C | | solubility | ≥50 mg/mL in DMSO; insoluble in H2O; ≥90.8 mg/mL in EtOH with gentle warming | | pka | 3.51±0.10(Predicted) | | form | solid | | color | White to off-white |
| WGK Germany | WGK 3 | | Storage Class | 11 - Combustible Solids |
| | Firategrast Usage And Synthesis |
| Uses | Treatment of multiple sclerosis and inflammatory bowel disease. | | Biological Activity | Firategrast is an orally available, potent and selective antagonist of α4β1/α4β7 integrins. Firategrast inhibits α4β1/α4β7 integrin-mediated transferring of lymphocytes into the central nervous system (CNS). In phase II trial it demonstrated a reduction of new contrast enhancing MRI lesions in patients with relapsing remitting MS. | | in vivo | Firategrast (30mg/kg/day in drinking water; starting 2 or 7 days post transplantation to 21 days) shows an overall reduction of leukemic cells in the spleen[3].
| Animal Model: | Female Wild-type C57BL/6J mice (8-12 weeks) with primary TCL1-tg splenocytes[3] | | Dosage: | 30mg/kg | | Administration: | Drinking water; daily; starting 2 or 7 days post transplantation to 21 days | | Result: | Showed an overall reduction of leukemic cells in the spleen, accompanied by significant spleen weight reduction.
|
| | target | α4β1/α4β7 integrin | | IC 50 | α4β1; α4β7 |
| | Firategrast Preparation Products And Raw materials |
|