| Company Name: |
Quality Control Solutions Ltd.
|
| Tel: |
0755-66853366 13670046396 |
| Email: |
orders@qcsrm.com |
| Products Intro: |
Product Name:Tenofovir Impurity 182
CAS:161760-03-8
Purity:95% HPLC Package:10mg;25mg;50mg;100mg
|
| Company Name: |
Nanjing XiZe Biotechnology CO., Ltd.
|
| Tel: |
025-66023220 15250997978 |
| Email: |
1511893459@qq.com |
| Products Intro: |
Product Name:Tenofovir Impurity 37
CAS:161760-03-8
Purity:95% HPLC Package:10mg;25mg;50mg;100mg;250mg;500mg;1g
|
Tenofovir Impurity 37 manufacturers
- Tenofovir Impurity 37
-

- $1.00 / 1g
-
2019-12-31
- CAS:161760-03-8
- Min. Order: 1g
- Purity: 99%
- Supply Ability: 1kg
|
| | Tenofovir Impurity 37 Basic information |
| Product Name: | Tenofovir Impurity 37 | | Synonyms: | Tenofovir Impurity 37;Ethyl p-toluenesulfonyloxymethylphosphonate;Phosphonic acid, [[[(4-methylphenyl)sulfonyl]oxy]methyl]-, monoethyl ester (9CI);Tenofovir Impurity 182;(ethoxy(hydroxy)phosphoryl)methyl 4-methylbenzenesulfonate;Tenofovir Impurity Z;(ethoxy(hydroxy)phosphoryl)methyl 4-methylbenzenesulfonate? (Tenofovir Impurity) | | CAS: | 161760-03-8 | | MF: | C10H15O6PS | | MW: | 294.26 | | EINECS: | | | Product Categories: | | | Mol File: | 161760-03-8.mol | 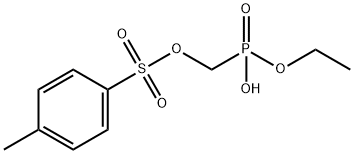 |
| | Tenofovir Impurity 37 Chemical Properties |
| Boiling point | 448.8±47.0 °C(Predicted) | | density | 1.369±0.06 g/cm3(Predicted) | | pka | 2.09±0.10(Predicted) |
| | Tenofovir Impurity 37 Usage And Synthesis |
| | Tenofovir Impurity 37 Preparation Products And Raw materials |
|