|
|
| | Z-DL-LYS(Z)-OH Basic information |
| Product Name: | Z-DL-LYS(Z)-OH | | Synonyms: | N-ALPHA,N-EPSILON-DI-Z-DL-LYSINE;N-ALPHA-N-EPSILON-DICARBOBENZOXY-DL-LYSINE;N-ALPHA,EPSILON-BIS-Z-DL-LYSINE;Z-DL-LYS(D)-OH;Z-DL-LYSINE(Z)-OH;Z-DL-LYS(Z)-OH;nα,nε-di-z-dl-lysine;N,N''-DICARBOBENZYLOXY-DL-LYSINE) | | CAS: | 55592-85-3 | | MF: | C22H26N2O6 | | MW: | 414.45 | | EINECS: | | | Product Categories: | | | Mol File: | 55592-85-3.mol | 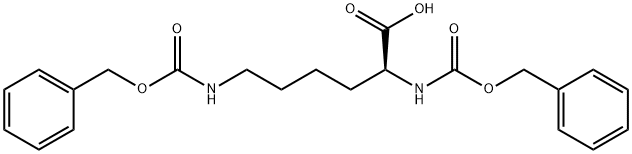 |
| | Z-DL-LYS(Z)-OH Chemical Properties |
| Melting point | 90-95 °C (lit.) | | Boiling point | 659.0±55.0 °C(Predicted) | | density | 1.238±0.06 g/cm3(Predicted) | | storage temp. | Sealed in dry,2-8°C | | pka | 3.97±0.21(Predicted) | | form | solid | | Appearance | White to off-white Solid | | BRN | 2027663 | | Major Application | peptide synthesis | | InChI | 1S/C22H26N2O6/c25-20(26)19(24-22(28)30-16-18-11-5-2-6-12-18)13-7-8-14-23-21(27)29-15-17-9-3-1-4-10-17/h1-6,9-12,19H,7-8,13-16H2,(H,23,27)(H,24,28)(H,25,26) | | InChIKey | BLZXFNUZFTZCFD-UHFFFAOYSA-N | | SMILES | OC(=O)C(CCCCNC(=O)OCc1ccccc1)NC(=O)OCc2ccccc2 |
| Safety Statements | 22-24/25 | | WGK Germany | 3 | | Storage Class | 11 - Combustible Solids |
| | Z-DL-LYS(Z)-OH Usage And Synthesis |
| Uses | peptide synthesis | | reaction suitability | reaction type: solution phase peptide synthesis | | Synthesis | GENERAL METHOD: 2-Amino-6-(((benzyloxy)carbonyl)amino)hexanoic acid (5.0 g, 30 mmol, 1.0 eq.) was dissolved in a solution of NaOH (2M, 50 mL) and stirred at 0°C and under nitrogen protection. Benzyl chloroformate (4.3 mL, 30 mmol, 1.0 eq.) was dissolved in toluene (5 mL) followed by addition of NaOH (4M, 5 mL) solution. The reaction mixture was stirred vigorously at room temperature for 30 minutes. Upon completion of the reaction, it was diluted with water (50 mL) and extracted with diethyl ether (2 x 50 mL). After combining the organic phases, it was cooled to 0°C and acidified with concentrated hydrochloric acid to pH 1, at which point a white solid precipitated. The solid product was collected by filtration. The resulting solid was dissolved in dichloromethane (200 mL), and the organic layer was washed with brine (50 mL), followed by drying with anhydrous magnesium sulfate, and finally concentrated under vacuum to obtain the target product Cbz-N'-Cbz-DL-lysine. | | References | [1] European Journal of Medicinal Chemistry, 2014, vol. 84, # C, p. 584 - 594 |
| | Z-DL-LYS(Z)-OH Preparation Products And Raw materials |
|