|
|
| | 4-MORPHOLIN-4-YL-BUTAN-1-OL Basic information |
| Product Name: | 4-MORPHOLIN-4-YL-BUTAN-1-OL | | Synonyms: | 4-Morpholinobutan-1-ol;AKOS BB/0085;4-MORPHOLIN-4-YL-BUTAN-1-OL;CHEMBRDG-BB 4001148;AKOS BBB/436;4-morpholin-4-ylbutan-1-ol(SALTDATA: FREE);4-Morpholino-1-butanol;4-Morpholinebutanol | | CAS: | 5835-79-0 | | MF: | C8H17NO2 | | MW: | 159.23 | | EINECS: | | | Product Categories: | | | Mol File: | 5835-79-0.mol | 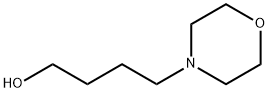 |
| | 4-MORPHOLIN-4-YL-BUTAN-1-OL Chemical Properties |
| Hazard Codes | Xi | | HazardClass | IRRITANT | | HS Code | 2934999090 |
| | 4-MORPHOLIN-4-YL-BUTAN-1-OL Usage And Synthesis |
| Synthesis | Example 3; 1-(4-Hydroxybutyl)morpholine was synthesized as follows: to a 250 mL three-necked flask were sequentially added morpholine (65.5 g, 750 mmol), 4-chloro-1-butanol (27.2 g, 250 mmol), sodium iodide (3.8 g, 25 mmol) and 75 mL of dioxane. The reaction mixture was heated to reflux for 48 hours. After completion of the reaction, the mixture was cooled to room temperature. The solvent was removed by rotary evaporator to give a brown oily crude product. The crude product was dissolved in 100 mL of 2N sodium hydroxide solution, extracted with ethyl acetate (EtOAc), the organic phases were combined and dried over anhydrous sodium sulfate (Na2SO4). After removal of the desiccant by filtration, the solvent was removed by distillation under reduced pressure to give an orange colored oil. Finally, the oily substance was subjected to reduced pressure distillation (90 °C, 1 mmHg) to collect the target product 1-(4-hydroxybutyl)morpholine (13.9 g, 87 mmol, 35% yield) as a colorless oil. | | References | [1] Patent: WO2008/66874, 2008, A1. Location in patent: Page/Page column 74-75
[2] Journal of the American Chemical Society, 1939, vol. 61, p. 3439
[3] Journal of the American Chemical Society, 1942, vol. 64, p. 970,972 |
| | 4-MORPHOLIN-4-YL-BUTAN-1-OL Preparation Products And Raw materials |
|