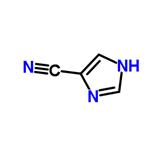
- 1H-Imidazole-4-carbonitrile
-
- $200.00 / 1KG
-
2025-09-25
- CAS:57090-88-7
- Min. Order: 1KG
- Purity: 99%, 99.5% Sublimated
- Supply Ability: g-kg-tons, free sample is available
|
| | 1H-Imidazole-4-carbonitrile Basic information |
| | 1H-Imidazole-4-carbonitrile Chemical Properties |
| Melting point | 143.5-144.5 °C | | Boiling point | 388.1±15.0 °C(Predicted) | | density | 1.28±0.1 g/cm3(Predicted) | | storage temp. | 2-8°C | | form | solid | | pka | 9.17±0.10(Predicted) | | color | Off-white to light yellow | | InChI | InChI=1S/C4H3N3/c5-1-4-2-6-3-7-4/h2-3H,(H,6,7) | | InChIKey | NWVGXXPWOYZODV-UHFFFAOYSA-N | | SMILES | C1NC(C#N)=CN=1 |
| Hazard Codes | Xi | | Risk Statements | 36 | | Safety Statements | 26 | | HazardClass | IRRITANT | | HS Code | 29339900 |
| | 1H-Imidazole-4-carbonitrile Usage And Synthesis |
| Uses | 1H-Imidazole-4-carbonitrile acts as a reagent in the structure-based optimization of a potent class of arylamide FMS inhibitors. | | Synthesis Reference(s) | The Journal of Organic Chemistry, 51, p. 3228, 1986 DOI: 10.1021/jo00366a037
Synthesis, p. 677, 2003 | | Synthesis | Example 72: General procedure for the synthesis of the potassium salt of 4-cyano-1-(2-trimethylsilyl-ethoxymethyl)-1H-imidazole-2-carboxylate
a) Synthesis of 1H-imidazole-4-carbonitrile
In a 22-L four-necked round-bottomed flask equipped with a mechanical stirrer, temperature probe, condenser, and charging funnel with nitrogen inlet, 1H-imidazole-4-carboxaldehyde (Aldrich, 1.10 kg, 11.5 mol) and pyridine (3.0 L, 3.0 mol) were added. The reaction flask was cooled to 8°C using an ice bath and hydroxylamine hydrochloride (871 g, 12.5 mol) was added slowly in batches, keeping the internal temperature below 30°C. The reaction mixture was cooled to ambient temperature and stirred at that temperature for 2 hours. Subsequently, the resulting thick yellow solution was heated to 80°C with a heating jacket and acetic anhydride (2.04 L, 21.6 mol) was added dropwise over a period of 200 min, keeping the temperature below 110°C during the dropwise addition. The reaction mixture was heated at 100 °C for 30 min and then cooled to ambient temperature and further cooled in an ice bath. The pH was adjusted to 8.0 (monitored using a pH meter) by adding 25 wt% NaOH solution (5.5 L) while maintaining the internal temperature below 30°C. The reaction mixture was transferred to a 22-L split funnel and extracted with ethyl acetate (6.0 L). The combined organic layers were washed with brine (2 x 4.0 L), dried over MgSO4, filtered, and concentrated to dryness under reduced pressure at 35 °C to give the crude product as a yellow semi-solid. The semi-solid was suspended in toluene (3.0 L), stirred for 1 h and filtered to give a light yellow solid. The solid was again suspended in toluene (3.0 L), stirred for 1 hour and filtered. The resulting slurry was filtered and the cake was washed with toluene (2 x 500 mL) to give the title compound as a light yellow solid (870 g, 82% yield).1H and 13C NMR spectra were consistent with the target structure. | | References | [1] Patent: WO2006/47504, 2006, A1. Location in patent: Page/Page column 146-147
[2] Patent: US2006/281788, 2006, A1. Location in patent: Page/Page column 88-89
[3] Patent: US2008/51402, 2008, A1. Location in patent: Page/Page column 74-75
[4] Patent: WO2007/48088, 2007, A2. Location in patent: Page/Page column 146-147
[5] Synthesis, 2008, # 21, p. 3377 - 3379 |
| | 1H-Imidazole-4-carbonitrile Preparation Products And Raw materials |
|