- Hydrocortisone
-

- $0.00 / 25kg
-
2025-12-01
- CAS:74050-20-7
- Min. Order: 1kg
- Purity: 98%
- Supply Ability: 1000kgs
|
| | Hydrocortisone aceponate Basic information |
| Product Name: | Hydrocortisone aceponate | | Synonyms: | hydrocortisone aceponate;[(8S,9S,10R,11S,13S,14S,17R)-17-(2-acetyloxyacetyl)-11-hydroxy-10,13-dimethyl-3-oxo-2,6,7,8,9,11,12,14,15,16-decahydro-1H-cyclopenta[a]phenanthren-17-yl] propanoate;[(8S,9S,10R,11S,13S,14S,17R)-17-(2-acetyloxyethanoyl)-11-hydroxy-10,13-dimethyl-3-oxo-2,6,7,8,9,11,12,14,15,16-decahydro-1H-cyclopenta[a]phenanthren-17-yl] propanoate;propionic acid [(8S,9S,10R,11S,13S,14S,17R)-17-(2-acetoxyacetyl)-11-hydroxy-3-keto-10,13-dimethyl-2,6,7,8,9,11,12,14,15,16-decahydro-1H-cyclopenta[a]phenanthren-17-yl] ester;Hydrocortisone 17-Propionate 21-Acetate;Hydrocortisonepropylacetate;Pregn-4-ene-3,20-dione, 21-(acetyloxy)-11-hydroxy-17-(1-oxopropoxy)-, (11β)-;Efficort | | CAS: | 74050-20-7 | | MF: | C26H36O7 | | MW: | 460.56 | | EINECS: | | | Product Categories: | Intermediates & Fine Chemicals;Pharmaceuticals;Steroids;API | | Mol File: | 74050-20-7.mol | 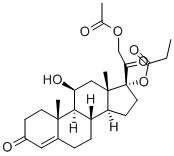 |
| | Hydrocortisone aceponate Chemical Properties |
| Melting point | 144-146°C | | density | 1.23±0.1 g/cm3 (20 ºC 760 Torr) | | storage temp. | Refrigerator | | solubility | Chloroform (Slightly), Methanol (Slightly) | | form | Solid | | color | White to Pale Beige | | InChIKey | MFBMYAOAMQLLPK-VRVFYZPPNA-N | | SMILES | [C@@]1(OC(=O)CC)(C(=O)COC(=O)C)CC[C@@]2([H])[C@]3([H])CCC4=CC(=O)CC[C@]4(C)[C@@]3([H])[C@@H](O)C[C@]12C |&1:0,15,17,27,29,31,34,r| |
| | Hydrocortisone aceponate Usage And Synthesis |
| Description | Hydrocortisone aceponate is the 17-a-propionate derivative of hydrocortisone-21-acetate.
Its is reportedly significantly superior to the latter in the treatment of dermatoses of
varying severity. | | Chemical Properties | White to Off-White Solid | | Originator | Beiersdorf (W.Germany) | | Uses | A new topical hydrocortisone derivative with esterification in positions 17 and 21, inhibit the incorporation of [3H]thymidine in DNA in human skin fibroblasts less strongly than the halogenated glucocorticosteroids betamethasone 17-valerate and clobetasol 17-propionate. Glucocorticosteroid. | | Definition | ChEBI: Hydrocortisone aceponate is a corticosteroid hormone. | | Brand name | Retef | | Synthesis | Hydrocortisone was prepared as follows: 1 8 g of hydrocortisone was dissolved in 18 ml of anhydrous dimethylformamide and treated with 16 ml of triethyl orthopropionate. After addition of 60 mg of p-toluenesulfonic acid 1H 2 O, the reaction mixture was stirred at about 11000 for 6 hours. After cooling to room temperature, 0.5 ml of pyridine was added and then the mixture was concentrated under vacuum. To the viscous residue dissolved in 1.2 liters of methanol was added a buffer mixture of 121.6 ml of 0.1 M trisodium citrate solution and 245 ml of 0.1 N hydrochloric acid and the mixture was boiled at reflux for 1 hour. After cooling the solution, the methanol was essentially removed under reduced pressure and the remaining suspension was treated with the same volume of water. The crystalline crude product precipitated by cooling was separated on a silica gel column using chloroform/ 3% methanol graduation. 9.5 g of pure acetylpropylhydrocortisone was obtained. Melting point: 192--1930C. |
| | Hydrocortisone aceponate Preparation Products And Raw materials |
|