2-(2-Aminothiazol-4-yl)glyoxylic acid manufacturers
|
| | 2-(2-Aminothiazol-4-yl)glyoxylic acid Basic information |
| Product Name: | 2-(2-Aminothiazol-4-yl)glyoxylic acid | | Synonyms: | 2-(2-AMINOTHIAZOL-4-YL) GLYOXYLIC ACID;2-(2-AMINOTHIAZOL-4-YL)-2-OXO-ACETIC ACID;2-amino-α-oxothiazol-4-acetic acid;2-amino-alpha-oxothiazol-4-acetic acid;ATGA:2-(2-AMINOTHIAZOL-4-YL) GLYOXYLIC ACID;2-(2-aminothiazol-4-yl)glyoxylic Acid (ATGA);ATGA;2-Amino-4-thiazoleglyoxylic Acid | | CAS: | 73150-67-1 | | MF: | C5H4N2O3S | | MW: | 172.16 | | EINECS: | 277-301-0 | | Product Categories: | Organic acids | | Mol File: | 73150-67-1.mol | 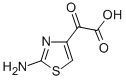 |
| | 2-(2-Aminothiazol-4-yl)glyoxylic acid Chemical Properties |
| Boiling point | 395.7±24.0 °C(Predicted) | | density | 1.711±0.06 g/cm3(Predicted) | | storage temp. | 2-8°C(protect from light) | | solubility | DMSO | | form | Solid | | pka | 1.25±0.54(Predicted) | | color | Pale Yellow | | InChI | InChI=1S/C5H4N2O3S/c6-5-7-2(1-11-5)3(8)4(9)10/h1H,(H2,6,7)(H,9,10) | | InChIKey | VMASTYPGLHRVNL-UHFFFAOYSA-N | | SMILES | C1(C(=O)C(=O)O)N=C(N)SC=1 | | CAS DataBase Reference | 73150-67-1(CAS DataBase Reference) |
| | 2-(2-Aminothiazol-4-yl)glyoxylic acid Usage And Synthesis |
| Uses | 2-Amino-4-thiazoleglyoxylic Acid is an intermediate in synthesizing (6R,7R)-7-(2-(2-Aminothiazol-4-yl)-2-oxoacetamido)-8-oxo-3-vinyl-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic Acid (A630310), which is used in the preparation of Cephalosporin C (C258750), an antibiotic used to study the effect of transpeptidase expression, binding, and inhibition on bacterial cell wall mucopeptide synthesis. | | Synthesis | Under ice bath cooling conditions, 10.1 g of methyl 2-(2-aminothiazol-4-yl)thioacetoxy S-acid (CAS: 89721-50-6) was mixed with 80 mL of water, followed by the addition of 10.6 g of sodium carbonate. The reaction mixture was stirred continuously for 1 h at a maintained ice bath temperature. Upon completion of the reaction, the pH of the reaction mixture was adjusted to 2.5 at the same temperature using 6 N hydrochloric acid. the precipitated crystals were collected by filtration, washed with deionized water and subsequently dried to afford 6.2 g (67.8% yield) of 2-(2-aminothiazol-4-yl)glyoxalic acid with a melting point of more than 200°C. The infrared spectra of the product (KBr pressed slice) showed a νC=O absorption peak at 1660 cm1. | | References | [1] Patent: US4563534, 1986, A |
| | 2-(2-Aminothiazol-4-yl)glyoxylic acid Preparation Products And Raw materials |
|