|
|
| | 1,3-BENZODITHIOLYLIUM TETRAFLUOROBORATE Basic information |
| Product Name: | 1,3-BENZODITHIOLYLIUM TETRAFLUOROBORATE | | Synonyms: | 1,3-BENZODITHIOL-2-YLIUM TETRAFLUOROBORATE;1,3-BENZODITHIOLYLIUM TETRAFLUOROBORATE;1,3-Benzodithiolylium tetrafluoroborate[Hydroxyl protecting agent];1,3-Benzodithiol-1-ium tetrafluoroborate 98%;bdtf;1,3-BENZODITHIOLYLIUM TETRAFLUOROBORATE 97%;1,3-Benzolithiolylium Tetrafluoroborate;Benzo[d][1,3]dithiol-1-ium tetrafluoroborate, BDTF | | CAS: | 57842-27-0 | | MF: | C7H5BF4S2 | | MW: | 240.05 | | EINECS: | | | Product Categories: | Heterocyclic Compounds;Iodonium Sulfonium & Oxonium Compounds;Sulfonium Compounds;B (Classes of Boron Compounds);Biochemistry;Nucleosides, Nucleotides & Related Reagents;Protecting Agents for Hydroxyl and Amino Groups;Protecting Agents, Phosphorylating Agents & Condensing Agents;Protection & Derivatization Reagents (for Synthesis);Sulfur Compounds (for Synthesis);Synthetic Organic Chemistry;Tetrafluoroborates | | Mol File: | 57842-27-0.mol | 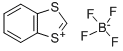 |
| | 1,3-BENZODITHIOLYLIUM TETRAFLUOROBORATE Chemical Properties |
| Melting point | 150 °C (dec.) (lit.) | | storage temp. | Inert atmosphere,Room Temperature | | solubility | sol MeCN and DMF (slowly reacts); insol ether. | | form | Solid | | color | Gray to brown | | BRN | 4731453 | | InChI | 1S/C7H5S2.BF4/c1-2-4-7-6(3-1)8-5-9-7;2-1(3,4)5/h1-5H;/q+1;-1 | | InChIKey | CUSWPJQKCZMDPY-UHFFFAOYSA-N | | SMILES | F[B-](F)(F)F.c1ccc2[s+]csc2c1 | | CAS DataBase Reference | 57842-27-0(CAS DataBase Reference) |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 26-36 | | RIDADR | 1759 | | WGK Germany | 3 | | F | 8-10-21 | | Hazard Note | Irritant | | HazardClass | 8 | | PackingGroup | III | | HS Code | 2934999090 | | Storage Class | 11 - Combustible Solids | | Hazard Classifications | Eye Irrit. 2
Skin Irrit. 2
STOT SE 3 |
| | 1,3-BENZODITHIOLYLIUM TETRAFLUOROBORATE Usage And Synthesis |
| Uses | 1,3-Benzodithiolylium tetrafluoroborate may be used in the synthesis of the following:
- Dibenzotetrathiafulvalene by reacting with 1,8-diazabicyclo[5.4.0]undec-7-ene.
- Substituted arylcarbenium ions by reacting with boronic derivatives.
- α-(1,3-benzodithiol-2-ylidene) ketones and 1,3-benzodithiole by reacting with ketones.
- 2-styryl-1,3-benzodithioles by reacting with styryl-type cobaloximes.
- 2-(3-indolyl)-1,3-benzodithiolylium tetrafluoroborate by reacting with indole.
- 2-(1,3-benzodithiol-2-ylidene)-3,5-cyclohexadien-1-ones and 4-(1,3-benzodithiol-2-ylidene)-2,5-cyclohexadien-1-ones by reacting with 2,4- and 2,6-disubstituted phenols.
| | Preparation | Preparative Method of 1,3-Benzodithiolylium tetrafluoroborate: treatment of 2-(3-methylbutoxy)-1,3-benzodithiole with Tetrafluoroboric Acid in
Acetic Anhydride at 0 °C. | | General Description | 1,3-Benzodithiolylium tetrafluoroborate is a carbenium ionic compound. It has been synthesized by reacting 1,3-benzodithiole with trityl tetrafluoroborate. It is widely used as an α-alkylating agent for the enantioselective α-alkylation of aldehydes., The nature of adsorption of 1,3-benzodithiolylium tetrafluoroborate on various carbon nanotubes (CNTs) has been studied. 1,3-Benzodithiolylium tetrafluoroborate reacts with different nucleophilic reagents at the 2-position to form the respective 1,3-benzodithioles. | | storage | 1,3-Benzodithiolylium tetrafluoroborate can be stored in the absence of moisture and light; no toxicity data is
currently available; 1,3-benzodithiol-1-ium perchlorate [32283-21-9] is also known, but poses a risk of
explosion. | | Purification Methods | 1,3-Benzodithiolylium tetrafluoroborate is recrystallized from acetic acid or acetonitrile/ether. |
| | 1,3-BENZODITHIOLYLIUM TETRAFLUOROBORATE Preparation Products And Raw materials |
|