- Valencene
-

- $113.00 / 500mg
-
2026-05-11
- CAS:4630-07-3
- Min. Order:
- Purity: 93.00%
- Supply Ability: 10g
- Valencene
-

- $0.00 / 1kg
-
2026-03-26
- CAS:4630-07-3
- Min. Order: 0.1kg
- Purity: 68.5%HPLC
- Supply Ability: 2000kgs
- VALENCENE
-

- $0.00 / 25KG
-
2026-03-20
- CAS:4630-07-3
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 100 MT
|
| | Valencene Basic information |
| Product Name: | Valencene | | Synonyms: | 7beta,8aalpha)]-theta-(1alph;Valencen;(3R,4AS,5R)-3-ISOPROPENYL-4A,5-DIMETHYL-1,2,3,4,4A,5,6,7-OCTAHYDRO-NAPHTHALENE;(3R,4AS,5R)-4A,5-DIMETHYL-3-ISOPROPENYL-1,2,3,4,4A,5,6,7-OCTAHYDRONAPHTHALENE;FEMA 3443;VALENCENE;VALENCENE 85;VALENCENE 65+% | | CAS: | 4630-07-3 | | MF: | C15H24 | | MW: | 204.35 | | EINECS: | 225-047-6 | | Product Categories: | Plant Oils, Toxins, Phenolic Acids & Derivatives | | Mol File: | 4630-07-3.mol | 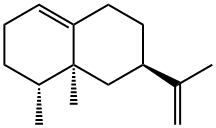 |
| | Valencene Chemical Properties |
| Boiling point | 274 °C(lit.) | | density | 0.92 g/mL at 25 °C(lit.) | | FEMA | 3443 | VALENCENE | | refractive index | n20/D 1.504(lit.) | | Fp | >230 °F | | storage temp. | 2-8°C | | solubility | Chloroform (Slightly), Ethyl Acetate (Slightly), Methanol (Slightly) | | form | Oil | | color | Colourless | | Specific Gravity | 0.92 | | Odor | at 10.00 % in dipropylene glycol. sweet fresh citrus grapefruit woody orange asprin dry green oily | | Odor Type | citrus | | Optical Rotation | [α]22/D +90°, neat | | biological source | oranges | | Hydrolytic Sensitivity | 1: no significant reaction with aqueous systems | | JECFA Number | 1337 | | BRN | 3539153 | | Henry's Law Constant | 3.2×10-4 mol/(m3Pa) at 25℃, Copolovici and Niinemets (2015) | | Stability: | Light Sensitive | | Major Application | cleaning products
cosmetics
flavors and fragrances
food and beverages
personal care | | Cosmetics Ingredients Functions | SKIN CONDITIONING
PERFUMING
SKIN PROTECTING | | InChI | 1S/C15H24/c1-11(2)13-8-9-14-7-5-6-12(3)15(14,4)10-13/h7,12-13H,1,5-6,8-10H2,2-4H3/t12-,13-,15+/m1/s1 | | InChIKey | QEBNYNLSCGVZOH-NFAWXSAZSA-N | | SMILES | C[C@@H]1CCC=C2CC[C@H](C[C@@]12C)C(C)=C | | LogP | 6.29 | | EPA Substance Registry System | Naphthalene, 1,2,3,5,6,7,8,8a-octahydro-1,8a-dimethyl-7-(1-methylethenyl)-, (1R,7R,8aS)- (4630-07-3) |
| Safety Statements | 23-24/25 | | WGK Germany | 3 | | TSCA | TSCA listed | | Storage Class | 10 - Combustible liquids | | Hazard Classifications | Asp. Tox. 1 |
| | Valencene Usage And Synthesis |
| Description | (+)-Valencene is a sesquiterpene that has been found in C. sativa and is an aromatic component of orange essence oil. (+)-Valencene (50 μM) induces heme oxgenase-1 (HO-1) expression in macrophages and inhibits the expression of inducible nitric oxide synthase (iNOS), the production of nitric oxide (NO), and the release of high-mobility group box-1 (HMGB1) in RAW 264.7 cells stimulated with LPS. It also increases the survival rate in a mouse model of sepsis induced by cecal ligation and puncture. (+)-Valencene has been used in the synthesis of nootkatone . | | Chemical Properties | clear colorless to yellowish liquid | | Occurrence | Reported found in citrus fruits, orange peel, orange juice, bitter orange peel oil, lemon peel oil, grapefruit
juice, grapefruit peel oil, kumquat peel oil, leaves and stalk of celery, clove stem, Thymus vulgaris L., fresh mango, globe artichoke,
cardamom, mangosteen and cocoa. | | Uses | Valencene is a sesquiterpene and an essential oil component found in a variety of plants, and has been shown to have functional antioxidant, antiradical and antimicrobial properties. | | Uses | (+)-Valencene can be used as a precursor to prepare:
- (+)-Nootkatone (a sesquiterpene) by dark singlet oxygenation.
- Benzoyloxyvalencene by reacting with tert-butyl peroxy benzoate via KharaschSosnovsky allylic oxidation method.
- (+)-Lineariifolianone, a natural product.
| | Definition | ChEBI: A carbobicyclic compound and sesquiterpene that is 1,2,3,4,4a,5,6,7-octahydronaphthalene which is substituted a prop-1-en-2-yl group at position 3 and by methyl groups at positions 4a and 5 (the 3R,4aS,5R
diastereoisomer). | | Preparation | By a Wolf–Kishner reduction of nootkatone. | | benefits | Strains high in Valencene produce euphoria and mood elevation, aid with alertness, and promote cognitive functions. Valencene is a powerful insecticide often found in tick and mosquito repellent. It is also used in pest control, cleaning, personal care, and cosmetics. | | General Description | Valencene is the major sesquiterpene aroma constituent of orange peel oil. It is mainly used as a starting material to synthesize nootkatone, an important flavor compound of grapefruit. | | References | [1] S. NAZ. Impact of Supercritical Fluid Extraction and Traditional Distillation on the Isolation of Aromatic Compounds from Cannabis indica and Cannabis sativa[J]. Journal of Essential Oil Bearing Plants, 2017, 106 1: 175-184. DOI: 10.1080/0972060x.2017.1281766
[2] áSLAUG HGNADóTTIR Russell L R. Identification of aroma active compounds in orange essence oil using gas chromatography–olfactometry and gas chromatography–mass spectrometry[J]. Journal of Chromatography A, 2003, 998 1: Pages 201-211. DOI: 10.1016/s0021-9673(03)00524-7
[3] KONSTANTIN TSOYI . (+)-Nootkatone and (+)-valencene from rhizomes of Cyperus rotundus increase survival rates in septic mice due to heme oxygenase-1 induction[J]. Journal of ethnopharmacology, 2011, 137 3: Pages 1311-1317. DOI: 10.1016/j.jep.2011.07.062
[4] JULIA KOLWEK. Cell-free one-pot conversion of (+)-valencene to (+)-nootkatone by a unique dye-decolorizing peroxidase combined with a laccase from Funalia trogii.[J]. Journal of Industrial Microbiology & Biotechnology, 2018, 45 2: 89-101. DOI: 10.1007/s10295-017-1998-9 |
| | Valencene Preparation Products And Raw materials |
|