Adagrasib manufacturers
- Adagrasib
-

- $0.00 / 1g
-
2026-04-24
- CAS:2326521-71-3
- Min. Order: 1g
- Purity: More Than 99%
- Supply Ability: 100kg/Month
- Adagrasib.
-

- $0.00 / 1g
-
2026-04-24
- CAS:2326521-71-3
- Min. Order: 1g
- Purity: More Than 99%
- Supply Ability: 100kg/Month
- MRTX-849
-

- $0.00 / 1gram
-
2025-12-25
- CAS:2326521-71-3
- Min. Order: 1gram
- Purity: 99%
- Supply Ability: 10kg
|
| | Adagrasib Basic information |
| Product Name: | Adagrasib | | Synonyms: | MRTX-849;MRTX849;MRTX-849;MRTX 849;2-PIPERAZINEACETONITRILE, 4-[7-(8-CHLORO-1-NAPHTHALENYL)-5,6,7,8-TETRAHYDRO-2-[[(2S)-1-METHYL-2-PYRR;2-Piperazineacetonitrile, 4-[7-(8-chloro-1-naphthalenyl)-5,6,7,8-tetrahydro-2-[[(2S)-1-methyl-2-pyrrolidinyl]methoxy]pyrido[3,4-d]pyrimidin-4-yl]-1-(2-fluoro-1-oxo-2-propen-1-yl)-, (2S)-;2-[(2S)-4-[7-(8-chloronaphthalen-1-yl)-2-[[(2S)-1-methylpyrrolidin-2-yl]methoxy]-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-4-yl]-1-(2-fluoroprop-2-enoyl)piperazin-2-yl]acetonitrile;MRTX849(Adagrasib);Adagrasib/MRTX-849;Adagrasib | | CAS: | 2326521-71-3 | | MF: | C32H35ClFN7O2 | | MW: | 604.12 | | EINECS: | | | Product Categories: | api;2326521-71-3 | | Mol File: | 2326521-71-3.mol | 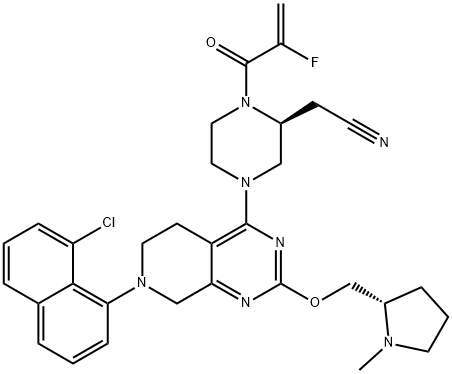 |
| | Adagrasib Chemical Properties |
| Boiling point | 860.2±75.0 °C(Predicted) | | density | 1.295±0.06 g/cm3(Predicted) | | storage temp. | Store at -20°C | | solubility | Soluble in DMSO:1.0(Max Conc. mg/mL);1.7(Max Conc. mM) | | pka | 9.31±0.40(Predicted) | | form | A crystalline solid | | color | White to yellow | | InChIKey | PEMUGDMSUDYLHU-ZEQRLZLVSA-N | | SMILES | N1(C(=O)C(F)=C)CCN(C2N=C(OC[C@@H]3CCCN3C)N=C3CN(C4=C5C(C=CC=C5Cl)=CC=C4)CCC3=2)C[C@@H]1CC#N |
| | Adagrasib Usage And Synthesis |
| Description |
MRTX-849 was identified as a potent, selective, and covalent KRASG12C inhibitor that exhibits favourable drug-like properties, modifies mutant cysteine 12 in GDP-bound KRASG12C, and inhibits KRAS-dependent signalling. MRTX-849 demonstrated pronounced tumour regression in 17 of 26 (65%) KRASG12C-positive cell line- and patient-derived xenograft models from multiple tumour types, and objective responses have been observed in patients with KRASG12C-positive lung and colon adenocarcinomas[1].
| | Uses | MRTX 849 is used as combination therapy for treatment of cancer. | | Definition | Adagrasib is an orally available, potent, irreversible, small molecule inhibitor of KRAS G12C mutant isoform being developed by Mirati Therapeutics for the treatment of solid tumours harbouring KRAS G12C oncogenic driver mutation, including non-small cell lung cancer (NSCLC) and colorectal cancer (CRC). | | Brand name | Krazati | | General Description | Class: non-kinase;
Treatment: KRAS(G12C) NSCLC;
Other name: MRTX849;
Elimination half-life: 24 h;
Protein binding = 98.3% | | Biological Activity |
To minimize GSH metabolism, the development candidate MRTX-849 employed a 2-fluoroacrylamide warhead, imparting whole blood stability of >50 h t1/2 across species while maintaining tractable cellular potency (IC50 = 5–14 nM). Metabolism studies of MRTX-849 in hepatocytes showed that GSH conjugation of the 2-fluoroacrylamide was reduced compared to previous analogues containing the unsubstituted acrylamide. Favorable in vitro ADME and physical properties of MRTX-849 translated into moderate to high bioavailability of 26–63% across species tested. In a PK/PD experiment dosed at 10, 30, and 100 mg/kg to NCI-H358 tumor-bearing mice, MRTX-849 demonstrated dose-dependent protein modification with nearly maximal effect at the highest doses[2].
| | Mechanism of action | Adagrasib selectively modified mutant cysteine 12 in GDP-bound G12C and inhibited KRASdependent signaling. | | Pharmacokinetics | The specifc KRAS G12C inhibitor MRTX849 inhibits the drug efux function of ABCB1 protein by interacting with the nucleotide binding domain of ABCB1, thereby increasing intracellular anticancer drug concentrations and overcoming MDR. While MRTX849 has attracted a lot of attention as a specifc KRAS G12C inhibitor for the treatment of KRAS-dependent tumors, this study revealed that MRTX849 may also be applied in combination with conventional chemotherapeutic agents for overcoming MDR. | | Clinical Use | Adagrasib is an irreversible inhibitor of the RAS GTPase family. It is approved by the FDA for the treatment of adult patients with locally advanced or metastatic non-small cell lung cancer (NSCLC) who have been identified by testing as having a KRAS (G12C) mutation. | | Synthesis | Boc deprotection was achieved by treatment with hydrochloric acid, and the resulting deprotected amine (19.5) crystallized as the tartrate salt in 80% isolated yield. Compound 19.5 was then coupled with naphthyl bromide 19.6 as the free base via the Buchwald-Hartwig reaction, and product 19.7 crystallized as the p-toluenesulfonate salt and was isolated in 71% yield. To avoid the complications caused by the potential mutagenic impurities and retro-Michael products that may be generated by the conventional carboxybenzoyl deprotection protocol, the authors developed a novel nucleophilic deprotection reaction using 2-mercaptoethanol and potassium phosphate in DMA. This approach afforded the deprotected piperazine 19.8 with minimal racemization, affording the crystalline product in 86% isolated yield. Finally, the cysteine-active warhead was introduced by coupling sodium 2-fluoroacrylate 19.9 with piperazine 19.8 using T3P (propylphosphonic anhydride) in acetonitrile, and Adagrasib (19) was isolated in 70% yield after two consecutive recrystallizations.

| | target | Primary target: KRAS(G12C) | | storage | Store at -20°C | | References |
[1] Jay B. Fell. “Identification of the Clinical Development Candidate MRTX849, a Covalent KRASG12C Inhibitor for the Treatment of Cancer.” Journal of Medicinal Chemistry 63 13 (2020): 6679–6693.
[2] J. Christensen. “The KRASG12C Inhibitor, MRTX849, Provides Insight Toward Therapeutic Susceptibility of KRAS Mutant Cancers in Mouse Models and Patients.” Cancer discovery (2019).
|
| | Adagrasib Preparation Products And Raw materials |
|