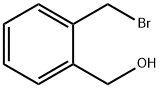
2-(BROMOMETHYL)BENZYL ALCOHOL, 95% synthesis
- Product Name:2-(BROMOMETHYL)BENZYL ALCOHOL, 95%
- CAS Number:74785-02-7
- Molecular formula:C8H9BrO
- Molecular Weight:201.06

612-14-6

74785-02-7
To 48% hydrobromic acid (15.0 mL) was added 1,2-benzenedimethanol (4.0 g, 29.0 mmol), and the reaction mixture was stirred for 2 h at room temperature. After the reaction was completed, 1N NaOH aqueous solution was added to neutralize to pH 7. The reaction mixture was diluted with ethyl acetate (200 mL), washed sequentially with saturated NaHCO3 solution (20 mL) and brine (20 mL), and the organic phase was dried over anhydrous Na2SO4. The solvent was removed by concentration under reduced pressure and the crude product was purified by silica gel column chromatography (Combi Flash Rf apparatus, ethyl acetate/hexane solvent system, 0-30% gradient elution) to afford 2.6 g (45% yield) of the target compound S14 as a white solid.1H NMR (500 MHz, CDCl3) δ: 7.30-7.45 (m, 4H), 4.85 (s, 2H), 4.64 (s, 2H), 1.81 (br s, 1H).

612-14-6
188 suppliers
$10.00/1g

74785-02-7
49 suppliers
$35.00/100mg
Yield:74785-02-7 56%
Reaction Conditions:
with hydrogen bromide in toluene at 70;
Steps:
4.9 2-(Bromomethyl)phenyl]methanol (10)
A solution of 105mg (0.76mmol, 1.00moleq) 1,2-phenylenedimethanol (9) in 20ml of toluene was heated to 70°C. At this temperature, 80μl (0.85mmol, 1.12moleq) of 48% solution of HBr was added dropwise. After 20min, TLC analysis confirmed a presence of product 10 and a small amount of probably bis brominated side product. Reaction mixture was cooled down, neutralized with saturated aq solution of Na2CO3 and extracted with diethyl ether (3×15ml). Combined organic layer was evaporated and resulted mixture purified by FLC (Hex / EA=5 / 1) to afford 80.0mg (0.40mmol, 56%) of desired product 10 as a white solid compound. Novelty: 2-(bromomethyl)phenyl]methanol (10) is described in the literature by its M.P., 1H NMR, IR22 and MS spectra.23 M.p.: 61.0-63.8°C [Et2O], lit: 55°C [hexane].22 (0035) 1H NMR (600MHz, CDCl3): δ 7.43 and 7.39 (2×dd, 2×1H, J(3,4 or 5,6)=7.5Hz, J(3,5 or 4,6)=1.5Hz, H-C(3 and 6)), 7.35 and 7.31 (2×ddd, 2×1H, J(4,5)=7.9Hz, J(3,4 or 5,6)=7.5Hz, J(3,5 or 4,6)=1.5Hz, H-C(4 and 5)), 4.86 and 4.66 (2×s, 2×2H, -CH2OH and CH2Br).
References:
Hlavá?, Matú?;Ková?iková, Lucia;?oltésová Prnová, Marta;Addová, Gabriela;Hanquet, Gilles;?tefek, Milan;Bohá?, Andrej [Bioorganic and Medicinal Chemistry,2021,vol. 29,art. no. 115885] Location in patent:supporting information

612-14-6
188 suppliers
$10.00/1g

74785-02-7
49 suppliers
$35.00/100mg

91-13-4
252 suppliers
$14.00/1g

85-44-9
781 suppliers
$9.00/5g

74785-02-7
49 suppliers
$35.00/100mg

643-79-8
745 suppliers
$9.00/1g

74785-02-7
49 suppliers
$35.00/100mg

88-99-3
480 suppliers
$15.00/50g

74785-02-7
49 suppliers
$35.00/100mg