Rosiglitazone maleate
- CAS No.
- 155141-29-0
- Chemical Name:
- Rosiglitazone maleate
- Synonyms
- Avandi;BRL-49653c;RSIGLITAZONEMALEATE;RosiglitazoneTartrate;ROSIGLITAZONE MALEATE;Rosiglitazone-d3 Maleate;Rosiglitazone Maleate forM;β-D-Fructopyranose1-Sulfamate;Rosiglitazone Maleate (200 mg);C16819000 RosiglitazonEMaleate
- CBNumber:
- CB0133276
- Molecular Formula:
- C22H23N3O7S
- Molecular Weight:
- 473.5
- MDL Number:
- MFCD03427306
- MOL File:
- 155141-29-0.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| Rosiglitazone maleate Pharmaceutical Secondary Standard; Certified Reference Material | PHR2513 | 300MG | $198 |
| Rosiglitazone - CAS 155141-29-0 - Calbiochem | 557366-M | 10MG | $131 |
| Rosiglitazone maleate | 1605817 | 200mg | $501 |
| Rosiglitazone Maleate | R0271 | 1G | $49 |
| Rosiglitazone Maleate | R0271 | 5G | $168 |
| More product size | |||
| Melting point | 235-240°C |
|---|---|
| storage temp. | 2-8°C |
| solubility | Soluble in DMSO (up to 50 mg/ml). |
| pka | 6.1; 6.8(at 25℃) |
| form | White powder |
| color | White |
| BCS Class | 1 |
| Stability | Stable for 2 years from date of purchase as supplied. Solutions in DMSO may be stored at -20°C for up to 3 months |
| Major Application | pharmaceutical (small molecule) |
| InChIKey | HCDYSWMAMRPMST-NTCAYCPXSA-N |
| SMILES | C(/C(=O)O)=C/C(=O)O.C(C1C=CC(OCCN(C2N=CC=CC=2)C)=CC=1)C1C(NC(=O)S1)=O |
| CAS DataBase Reference | 155141-29-0(CAS DataBase Reference) |
| FDA UNII | KX2339DP44 |
| NCI Dictionary of Cancer Terms | rosiglitazone maleate |
| NCI Drug Dictionary | rosiglitazone maleate |
| UNSPSC Code | 41116107 |
| NACRES | NA.77 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H315-H319 | |||||||||
| Precautionary statements | P264-P280-P302+P352-P337+P313-P305+P351+P338-P362+P364-P332+P313 | |||||||||
| Hazard Codes | Xi | |||||||||
| Risk Statements | 20/21/22-36/38 | |||||||||
| Safety Statements | 24/25-37/39-26 | |||||||||
| WGK Germany | 3 | |||||||||
| Storage Class | 11 - Combustible Solids | |||||||||
| NFPA 704 |
|
Rosiglitazone maleate price More Price(56)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | PHR2513 | Rosiglitazone maleate Pharmaceutical Secondary Standard; Certified Reference Material | 155141-29-0 | 300MG | $198 | 2026-04-30 | Buy |
| Sigma-Aldrich | 557366-M | Rosiglitazone - CAS 155141-29-0 - Calbiochem | 155141-29-0 | 10MG | $131 | 2026-04-30 | Buy |
| Sigma-Aldrich | 1605817 | Rosiglitazone maleate | 155141-29-0 | 200mg | $501 | 2026-04-30 | Buy |
| TCI Chemical | R0271 | Rosiglitazone Maleate | 155141-29-0 | 1G | $49 | 2026-04-30 | Buy |
| TCI Chemical | R0271 | Rosiglitazone Maleate | 155141-29-0 | 5G | $168 | 2026-04-30 | Buy |
Rosiglitazone maleate Chemical Properties,Uses,Production
Description
Rosiglitazone maleate, belongs to a novel class of thiazolidine diones launched for the treatment of non-insulin-dependent diabetes mellitus (NIDDM), a disease characterized by a pancreatic β-cell defect and insulin resistance in the liver and peripheral tissues. The racemic base can be obtained by KnSvenagel condensation between 2, 4-thiazolidinedione and the corresponding 4-substituted benzaldehyde (itself prepared in 2 steps from 2-chloropyridine), followed by reduction of the benzylidene. Rosiglitazone was shown to be a potent agonist of peroxisome proliferator activated receptor-gamma (PPARgamma), a nuclear receptor involved in the differentiation of adipose tissue, without activating liver PPAR-alpha receptors. This activation of PPAR-gamma could mediate the down-regulation of leptin gene expression. In animal models, Rosiglitazone has been shown to normalize glucose metabolism and reduce the exogenous dose of insulin needed to achieve glycemic control. In patients with Type II diabetes, daily doses (4 or 8 mg) of Rosiglitazone significantly improved blood sugar control without affecting cardiac structure or function.
Chemical Properties
White To Off-White Solid
Originator
SmithKline Beecham (US)
Uses
Insulin sensitizer; binds to peroxisome proliferator activated receptor gamma (PPAR- γ).
Uses
Thiazole alkanes antidiabetic drug
Uses
Thiazolidinediones (TZDs) are a group of structurally related peroxisome proliferator-activated receptor γ (PPARγ) agonists with antidiabetic actions in vivo. Rosiglitazone is a prototypical TZD that has served as a reference compound for this class. It is a potent and selective PPARγ ligand that binds to the PPARγ ligand-binding domain with a Kd value of 43 nM. It activates luciferase-based expression constructs PPARγ1 and PPARγ2 with EC50 values of approximately 30 nM and 100 nM, respectively. Rosiglitazone is active in vivo as an antidiabetic agent in the ob/ob mouse model, and has been used as an oral hypoglycemic agent in the treatment of type 2 diabetes in humans for many years.[Cayman Chemical]
Indications
Rosiglitazone maleate is an antihyperglycemic agent in the thiazolidinedione class. It is indicated for the treatment of patients with type 2 diabetes mellitus.
brand name
Avandia (GlaxoSmithKline).
General Description
A thiazolidinedione compound that acts as an anti-diabetic agent and serves as a potent and selective agonist of peroxisome proliferator-activated receptor-g (PPARg) (Kd ~40 nM) in fat cells. Shown to reduce fatty acid uptake and ameliorate lipid metabolism and insulin resistance in animal models of type II diabetes. Reported to activate both a1- and a2-containing AMPK complexes. Unlike troglitazone, it does not induce the activity of P4503A4. Significantly improves the differentiation of C3H10T1/2 stem cells into adipocytes. Shown to block estrogen synthesis by interfering with androgen binding to aromatase, but without affecting aromatase mRNA or protein expression.
Synthesis
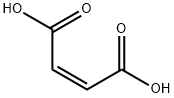
110-16-7

122320-73-4

155141-29-0
Example 3: Preparation of rosiglitazone maleate 93.3 g (261.0 mmol) of rosiglitazone base prepared in Example 2 and 36.4 g (313.2 mmol) of maleic acid were suspended in 510 mL of isopropanol. The suspension was heated to reflux temperature and held for 1 hour until the initial suspension was transformed into a clarified colorless solution. Subsequently, the solution was cooled to 10 °C and filtered using a Büchner funnel. The filter cake was washed with 200 mL of isopropanol to yield 105.0 g of crude rosiglitazone maleate (85% yield, 99.57% HPLC purity). 41.1 g (86.9 mmol) of the above crude solid was suspended with 1.7 g (14.5 mmol) of maleic acid in 200 mL of ethanol (the ratio of maleic acid to ethanol was about 13.5:1, i.e., 0.17 equivalents of maleic acid per 2.3 L of ethanol). The suspension was heated to reflux temperature and maintained for 20 minutes. After that, the solution was cooled to 65 °C and the crystal seed particles of the desired rosiglitazone maleate polymorph were added. The resulting suspension was further cooled to 0 °C and filtered using a Büchner funnel. The filter cake was washed with 50 mL of ethanol to give a final product of 39.5 g of rosiglitazone maleate (96% yield, 99.89% HPLC purity) with a melting point of 120.6-121.7°C.
storage
+4°C
Background
Rosiglitazone Maleate is an agonist of peroxisome proliferator-activated receptor γ, a ligand-activated nuclear receptor preferentially expressed in adipocytes, vascular smooth muscle cells, and macrophages. Rosiglitazone Maleate mediates adipogenesis, lipid metabolism, and insulin sensitivity. Rosiglitazone Maleate binds both PPARγ1 and PPARγ2 isoforms with high affinity. Treatment of mesenchymal stem cells with Rosiglitazone Maleate resulted in adipocyte differentiation. The treatment of type 2 diabetes mellitus using Rosiglitazone Maleate is controversial as its use can lead to serious side effects, including myocardial hypertrophy and heart failure. Exposure of murine neuroblastoma cells and primary hippocampal neurons to Rosiglitazone Maleate protected these cells from oxidative stress. Rosiglitazone Maleate binding to PPARγ activated transcription of neurotrophic factor-α1, which promoted increased expression of BCL-2 and inhibition of caspase-3. Studies suggest that Rosiglitazone Maleate prevents microglial activation, promotes microglial anti-inflammatory polarization, and suppresses inflammatory cytokines in inflammation-related diseases. In a mouse model of status epilepticus, Rosiglitazone Maleate prevented microglial activation and inhibited pilocarpine-induced status epilepticus.
References
[1] BARRIE C. C. CANTELLO. [[.omega.-(Heterocyclylamino)alkoxy]benzyl]-2,4-thiazolidinediones as potent antihyperglycemic agents[J]. Journal of Medicinal Chemistry, 1994, 37 23: 3977-3985. DOI:10.1021/jm00049a017
[2] J M LEHMANN. An antidiabetic thiazolidinedione is a high affinity ligand for peroxisome proliferator-activated receptor gamma (PPAR gamma).[J]. The Journal of Biological Chemistry, 1995, 270 22: 12953-12956. DOI:10.1074/jbc.270.22.12953
[3] TIMOTHY M. WILLSON. The Structure−Activity Relationship between Peroxisome Proliferator-Activated Receptor γ Agonism and the Antihyperglycemic Activity of Thiazolidinediones[J]. Journal of Medicinal Chemistry, 1996, 39 3: 665-668. DOI:10.1021/jm950395a
[4] HARUYA OHNO. PPARγ agonists induce a white-to-brown fat conversion through stabilization of PRDM16 protein.[J]. Cell metabolism, 2012: 395-404. DOI:10.1016/j.cmet.2012.01.019
[5] FRANCISCO JAVIER RUIZ-OJEDA. Cell Models and Their Application for Studying Adipogenic Differentiation in Relation to Obesity: A Review.[J]. International Journal of Molecular Sciences, 2016, 17 7. DOI:10.3390/ijms17071040
Rosiglitazone maleate Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| career henan chemical co | +86-0371-86658258 +8613203830695 | sales@coreychem.com | China | 29818 | 58 |
| Xiamen AmoyChem Co., Ltd | +86-86-5926051114 +8615060885618 | sales@amoychem.com | China | 6369 | 58 |
| HubeiwidelychemicaltechnologyCo.,Ltd | 18627774460 | faith@widelychemical.com | CHINA | 742 | 58 |
| Alchem Pharmtech,Inc. | 8485655694 | sales@alchempharmtech.com | United States | 63687 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | sales@conier.com | China | 49977 | 58 |
| TargetMol Chemicals Inc. | +1-781-999-5354; +17819995354 | marketing@targetmol.com | United States | 32467 | 58 |
| Hubei Ipure Biology Co., Ltd | +8613367258412 | ada@ipurechemical.com | China | 10236 | 58 |
| HANGZHOU CLAP TECHNOLOGY CO.,LTD | 86-571-88216897,88216896 13588875226 | sales@hzclap.com | CHINA | 6312 | 58 |
| ANHUI WITOP BIOTECH CO., LTD | +8615255079626 | eric@witopchemical.com | China | 23541 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-029-89586680 +86-18192503167 | 1026@dideu.com | China | 10000 | 58 |
View Lastest Price from Rosiglitazone maleate manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2026-05-06 | Rosiglitazone maleate
155141-29-0
|
US $0.00-0.00 / kg | 1kg | 98.0% | 1kg/month | WUHAN FORTUNA CHEMICAL CO., LTD | |
 |
2026-04-22 | Rosiglitazone maleate
155141-29-0
|
US $48.00-29.00 / mg | 99.76% | 10g | TargetMol Chemicals Inc. | ||
 |
2023-03-27 | Rosiglitazone maleate
155141-29-0
|
US $80.00 / KG | 1KG | 99.9% | 500MT/month | XINGTAI XINGJIU NEW MATERIAL TECHNOLOGY CO., LTD |
-

- Rosiglitazone maleate
155141-29-0
- US $0.00-0.00 / kg
- 98.0%
- WUHAN FORTUNA CHEMICAL CO., LTD
-

- Rosiglitazone maleate
155141-29-0
- US $48.00-29.00 / mg
- 99.76%
- TargetMol Chemicals Inc.
-

- Rosiglitazone maleate
155141-29-0
- US $80.00 / KG
- 99.9%
- XINGTAI XINGJIU NEW MATERIAL TECHNOLOGY CO., LTD





