|
|
| | 2,5-Dimethoxyphenylboronic acid Basic information |
| Product Name: | 2,5-Dimethoxyphenylboronic acid | | Synonyms: | 2,5-DIMETHOXYPHENYLBORONIC ACID,99+%;2,5-Dimethoxyphenylboronic Acid (contains varying amounts of Anhydride);2,5-Dimethoxyphenylboronic acid ,98%;2,5-Dimethoxyphenylboronic acid,97%;2,5-Dimethoxyphenylboronic acid, May contain varying amounts of anhydride, 97%;(2,5-dimethoxyphenyl)boronic acid(SALTDATA: FREE);AKOS BRN-0036;2,5-DIMETHOXYPHENYLBORONIC ACID | | CAS: | 107099-99-0 | | MF: | C8H11BO4 | | MW: | 181.98 | | EINECS: | 679-699-5 | | Product Categories: | Boronic Acid;Alkoxy;Aryl;Organoborons;B (Classes of Boron Compounds);Boronic Acids;Boronic Acids;Boronic Acids and Derivatives;BoronicAcids;Boric Acid;blocks | | Mol File: | 107099-99-0.mol | 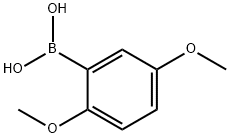 |
| | 2,5-Dimethoxyphenylboronic acid Chemical Properties |
| Melting point | 95-98 °C (lit.) | | Boiling point | 374.5±52.0 °C(Predicted) | | density | 1.19±0.1 g/cm3(Predicted) | | storage temp. | Keep in dark place,Sealed in dry,Room Temperature | | solubility | soluble in Methanol | | form | solid | | pka | 8.12±0.58(Predicted) | | color | White to Light yellow | | BRN | 2969872 | | InChI | InChI=1S/C8H11BO4/c1-12-6-3-4-8(13-2)7(5-6)9(10)11/h3-5,10-11H,1-2H3 | | InChIKey | QOZLFNQLIKOGDR-UHFFFAOYSA-N | | SMILES | B(C1=CC(OC)=CC=C1OC)(O)O | | CAS DataBase Reference | 107099-99-0(CAS DataBase Reference) |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 37/39-26-36/37-36/37/39 | | WGK Germany | 3 | | Hazard Note | Irritant/Keep Cold | | HazardClass | IRRITANT | | HS Code | 29319090 | | Storage Class | 11 - Combustible Solids |
| | 2,5-Dimethoxyphenylboronic acid Usage And Synthesis |
| Chemical Properties | white to almost white crystalline powder | | Uses | suzuki reaction | | Uses | 2,5-Dimethoxyphenylboronic acid is a bacterial mutagen. | | Synthesis | In a 1 L round bottom flask, 1-bromo-2,5-dimethoxybenzene (50 g, 230 mmol) was dissolved in tetrahydrofuran (400 ml). After cooling the reaction mixture to -78°C, n-butyllithium (167 ml, 280 mmol) was added slowly and dropwise. The reaction temperature was maintained at -78°C with continuous stirring for 2 hours. Subsequently, trimethyl borate (36 ml, 320 mmol) was added. The reaction mixture was gradually warmed to room temperature and stirred overnight. Upon completion of the reaction, acidification was carried out by slow dropwise addition of 2N HCl solution. Extraction was carried out with deionized water and ethyl acetate and the organic layer was separated and dried over anhydrous magnesium sulfate. The dried organic phase was concentrated in vacuum to give the crude product. Finally, the purified 2,5-dimethoxyphenylboronic acid (20.8 g, 50% yield) was obtained by recrystallization from a solvent mixture of heptane and toluene. | | References | [1] Patent: US2017/18723, 2017, A1. Location in patent: Paragraph 0157-0158
[2] Patent: US5688808, 1997, A
[3] Patent: US5693646, 1997, A
[4] Patent: US5688810, 1997, A
[5] Patent: US5696127, 1997, A |
| | 2,5-Dimethoxyphenylboronic acid Preparation Products And Raw materials |
|