2-BENZYLOXYPYRIDINE-5-BORONIC ACID, PINACOL ESTER manufacturers
|
| | 2-BENZYLOXYPYRIDINE-5-BORONIC ACID, PINACOL ESTER Basic information |
| Product Name: | 2-BENZYLOXYPYRIDINE-5-BORONIC ACID, PINACOL ESTER | | Synonyms: | 2-Benzyloxy-5-pyridineboronic acid pinacol ester;2-BENZYLOXY-5-(4,4,5,5-TETRAMETHYL-[1,3,2]-DIOXABOROLAN-2-YL)PYRIDINE;2-BENZYLOXYPYRIDINE-5-BORONIC ACID, PINACOL ESTER;6-(Benzyloxy)pyridine-3-boronic acid pinacol ester;Pyridine, 2-(phenylmethoxy)-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-;6-benzoxypyridine-3-borate frequency alcohol ester;6-benzyloxypyridine-3-boronic acidester | | CAS: | 832735-54-3 | | MF: | C18H22BNO3 | | MW: | 311.18 | | EINECS: | | | Product Categories: | Heterocyclic Compounds | | Mol File: | 832735-54-3.mol | 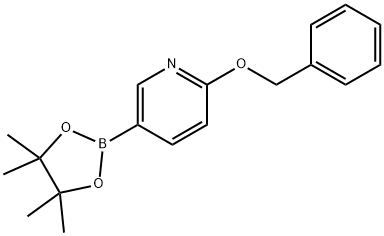 |
| | 2-BENZYLOXYPYRIDINE-5-BORONIC ACID, PINACOL ESTER Chemical Properties |
| Melting point | 138-142°C | | Boiling point | 429.6±35.0 °C(Predicted) | | density | 1.11±0.1 g/cm3(Predicted) | | storage temp. | 2-8°C | | form | solid | | pka | 3.91±0.24(Predicted) | | color | Yellow | | InChI | 1S/C18H22BNO3/c1-17(2)18(3,4)23-19(22-17)15-10-11-16(20-12-15)21-13-14-8-6-5-7-9-14/h5-12H,13H2,1-4H3 | | InChIKey | MXNLRVZITPPZHT-UHFFFAOYSA-N | | SMILES | CC1(C)OB(OC1(C)C)c2ccc(OCc3ccccc3)nc2 | | CAS DataBase Reference | 832735-54-3 |
| Risk Statements | 36/37/38 | | Safety Statements | 26-36/37/39 | | WGK Germany | 3 | | HS Code | 2933399990 | | Storage Class | 11 - Combustible Solids | | Hazard Classifications | Aquatic Chronic 4 |
| | 2-BENZYLOXYPYRIDINE-5-BORONIC ACID, PINACOL ESTER Usage And Synthesis |
| Synthesis | General procedure: to a 50 mL solution of 1,4-dioxane was added sequentially 2-benzyloxy-5-bromopyridine (3.76 g, 14.2 mmol, 1.0 eq.), pinacol ester of bis(boronic acid) (4.34 g, 17.1 mmol, 1.2 eq.), potassium acetate (3.48 g, 35.5 mmol, 2.5 eq.), and [1,1'-bis(diphenylphosphino) ferrocene]palladium dichloride (1.16 g, 1.4 mmol, 0.1 eq.). The reaction mixture was heated to 100 °C under nitrogen protection and refluxed overnight. After completion of the reaction, the reaction mixture was concentrated and dried, and the residue was purified by column chromatography (eluent: petroleum ether/dichloromethane=2:1) to afford 6-benzyloxypyridine-3-boronic acid pinacol ester (2.63 g, 59.5% yield). | | References | [1] Patent: CN106588937, 2017, A. Location in patent: Paragraph 0197 |
| | 2-BENZYLOXYPYRIDINE-5-BORONIC ACID, PINACOL ESTER Preparation Products And Raw materials |
|