- Benzomorpholine
-
- $0.00 / 1KG
-
2025-04-04
- CAS:5735-53-5
- Min. Order: 1KG
- Purity: 98%
- Supply Ability: 1Ton
- Benzomorpholine
-
- $0.00 / 1KG
-
2022-01-05
- CAS:5735-53-5
- Min. Order: 1g
- Purity: 98%min
- Supply Ability: 100kgs/month
|
| | Benzomorpholine Basic information |
| Product Name: | Benzomorpholine | | Synonyms: | 7-Oxa-10-azabicyclo[4.4.0]deca-1,3,5-triene;AKOS BC-1149;AKOS BBS-00002717;2,3-dihydro-1,4-benzoxazine;Benzomorpholine(3,4-Dihydro-2H-1,4-benzoxazine);3,4-DIHYDRO-2H-1,4-BENZOXAZINE;BENZOMORPHOLINE;CHEMBRDG-BB 4005008 | | CAS: | 5735-53-5 | | MF: | C8H9NO | | MW: | 135.16 | | EINECS: | | | Product Categories: | pharmacetical;Heterocycles series | | Mol File: | 5735-53-5.mol | 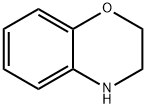 |
| | Benzomorpholine Chemical Properties |
| Boiling point | 85°C/0.75mm | | density | 1.095±0.06 g/cm3(Predicted) | | refractive index | 1.60 | | storage temp. | Keep in dark place,Inert atmosphere,Room temperature | | pka | 4.37±0.20(Predicted) | | form | clear liquid | | color | Colorless to Yellow to Orange | | Water Solubility | Sparingly soluble in water.(0.26 g/L) (25°C), | | InChI | InChI=1S/C8H9NO/c1-2-4-8-7(3-1)9-5-6-10-8/h1-4,9H,5-6H2 | | InChIKey | YRLORWPBJZEGBX-UHFFFAOYSA-N | | SMILES | O1C2=CC=CC=C2NCC1 |
| | Benzomorpholine Usage And Synthesis |
| Uses | It is used as pharmaceutical intermediate. Benzoylthiophenes are allosteric enhancers (AE) of agonist activity at the A1 adenosine receptor. | | Definition | ChEBI: 3,4-Dihydro-2H-1,4-benzoxazine is a benzoxazine. | | Synthesis | To a suspension of 2-aminophenol (1.0 g, 9.2 mmol) and potassium carbonate (6.36 g, 46 mmol) in anhydrous DMF (10 mL) was added 1,2-dibromoethane (2.59 g, 13.8 mmol). The reaction mixture was heated at 125 °C with stirring for 15 hours. After completion of the reaction, it was cooled to room temperature, the mixture was poured into crushed ice and extracted with ethyl acetate (3 × 20 mL). The organic phases were combined, dried with anhydrous sodium sulfate and concentrated under reduced pressure to remove the solvent. The resulting crude product was purified by silica gel column chromatography with ethyl acetate/petroleum ether (1:10, v/v) as eluent to afford 3,4-dihydro-2H-benzo[b][1,4]oxazine as a red oil (0.81 g, 65% yield). The product was analyzed by LC-MS (ES-API): retention time 7.51 min; calculated value C8H9NO [M+H]+ 136.1, measured value 136.1. | | References | [1] Tetrahedron Letters, 2006, vol. 47, # 44, p. 7823 - 7826
[2] Patent: WO2011/47432, 2011, A1. Location in patent: Page/Page column 88
[3] Heterocyclic Communications, 2012, vol. 18, # 3, p. 143 - 146
[4] Patent: WO2010/51374, 2010, A1. Location in patent: Page/Page column 132
[5] Patent: EP2172453, 2010, A1. Location in patent: Page/Page column 13 |
| | Benzomorpholine Preparation Products And Raw materials |
| Raw materials | 2-(2-nitrophenoxy)acetonitrile-->Benzene, 1-(2,2-diethoxyethoxy)-2-nitro--->1-(2,3-dihydro-1,4-benzoxazin-4-yl)ethanone-->2-(2-BroMo-phenylaMino)-ethanol-->Allyl 2-nitrophenyl ether-->1-(2-BROMOETHOXY)-2-NITROBENZENE-->2-(2-CHLOROETHOXY)NITROBENZENE-->2-(2-Amino-phenoxy)-ethanol-->3(2H)-benzofuranone, oxime, (3Z)--->4-Methyl-2,3-dihydro-1,4-benzoxazine-->2H-1,4-BENZOXAZIN-3(4H)-ONE-->2-Aminophenol-->1,2-Dibromoethane | | Preparation Products | 4-Chloro-3-hydroxybenzoyl chloride |
|