|
|
| | 1-(2-Fluoro-6-(trifluoromethyl)benzyl)-6-methylpyrimidine-2,4(1H,3H)-dione Basic information |
| Product Name: | 1-(2-Fluoro-6-(trifluoromethyl)benzyl)-6-methylpyrimidine-2,4(1H,3H)-dione | | Synonyms: | Benzeneacetic acid, 4-fluoro-.alpha.-(1-methylethyl)-, (.alpha.S)-;1-(2-Fluoro-6-trifluoromethyl-benzyl)-6-methyl-1H-pyrimidine-2,4-dione;1-[[2-fluoro-6-(trifluoromethyl)phenyl]methyl]-6-methylpyrimidine-2,4-dione;2,4(1H,3H)-Pyrimidinedione, 1-[[2-fluoro-6-(trifluoromethyl)phenyl]methyl]-6-methyl-;4-fluoro-.alpha.-(1-methylethyl)-, (.alpha.S)-;elagolix intermediate;elagolix intermediate 4;1-[2-Fluoro-6-(trifluoromethyl)benzyl]-6-methylpyrimidine-2,4(1H,3H)-dione | | CAS: | 830346-47-9 | | MF: | C13H10F4N2O2 | | MW: | 302.22 | | EINECS: | | | Product Categories: | 830346-47-9 | | Mol File: | 830346-47-9.mol | 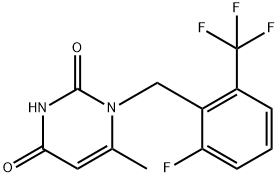 |
| | 1-(2-Fluoro-6-(trifluoromethyl)benzyl)-6-methylpyrimidine-2,4(1H,3H)-dione Chemical Properties |
| density | 1.413±0.06 g/cm3(Predicted) | | storage temp. | Sealed in dry,Room Temperature | | pka | 9.49±0.40(Predicted) | | Appearance | White to off-white Solid | | InChI | InChI=1S/C13H10F4N2O2/c1-7-5-11(20)18-12(21)19(7)6-8-9(13(15,16)17)3-2-4-10(8)14/h2-5H,6H2,1H3,(H,18,20,21) | | InChIKey | IJRKKZXPXBTHER-UHFFFAOYSA-N | | SMILES | C1(=O)N(CC2=C(C(F)(F)F)C=CC=C2F)C(C)=CC(=O)N1 |
| | 1-(2-Fluoro-6-(trifluoromethyl)benzyl)-6-methylpyrimidine-2,4(1H,3H)-dione Usage And Synthesis |
| Uses | 1-(2-Fluoro-6-(trifluoromethyl)benzyl)-6-methylpyrimidine-2,4(1H,3H)-dione is a pharmaceutical intermediate ingredient used in the preparation of Elagolix sodium, which is FDA-approved for the treatment of pain associated with moderate to severe pain associated with endometriosis. | | Synthesis | Using 1-(2-fluoro-6-(trifluoromethyl)benzyl)urea (2.568 g, 10.9 mmol) and tert-butyl acetoacetate (5.0 g) in toluene (125 mL), the mixture was briefly heated to reflux using a Dean-Stark splitter. Subsequently, tert-butyl acetoacetate (5.0 g) was added and heating to reflux was continued for 4 hours. Next, p-toluenesulfonic acid monohydrate (2.82 g, 14.8 mmol) was added and reflux was continued for 1 hour. Upon completion of the reaction, toluene was replaced with isopropanol (i-PrOH) and the volume of the solution was concentrated to approximately 30 mL. The solution was stirred overnight at room temperature to crystallize the product. The crystallized product was collected by filtration and washed with a small amount of isopropanol to give 2.01 g of 1-(2-fluoro-6-(trifluoromethyl)benzyl)-6-methylpyrimidine-2,4(1H,3H)-dione in 63% yield. The product was analyzed by LCMS (ESI) at m/z 303.0 (MH+). | | References | [1] Patent: WO2009/62087, 2009, A1. Location in patent: Page/Page column 8 |
| | 1-(2-Fluoro-6-(trifluoromethyl)benzyl)-6-methylpyrimidine-2,4(1H,3H)-dione Preparation Products And Raw materials |
|