|
|
| | 2-methoxypropionic acid Basic information |
| Product Name: | 2-methoxypropionic acid | | Synonyms: | 2-methoxypropanoic acid;Einecs 224-356-3;Propanoic acid, 2-methoxy-;2-methoxypropanoic acid(SALTDATA: FREE);2-methoxypropionic acid;alpha-Methoxypropionic acid;2-Methoxypropionsure;2021.11.2 | | CAS: | 4324-37-2 | | MF: | C4H8O3 | | MW: | 104.1 | | EINECS: | 224-356-3 | | Product Categories: | | | Mol File: | 4324-37-2.mol | 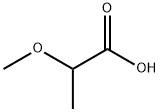 |
| | 2-methoxypropionic acid Chemical Properties |
| Melting point | 128.5 °C | | Boiling point | 199℃ | | density | 1.085 | | Fp | 87℃ | | storage temp. | 2-8°C | | solubility | Chloroform (Slightly), Methanol (Slightly) | | pka | 3.59±0.10(Predicted) | | form | Oil | | color | Colorless | | InChI | InChI=1S/C4H8O3/c1-3(7-2)4(5)6/h3H,1-2H3,(H,5,6) | | InChIKey | ICPWFHKNYYRBSZ-UHFFFAOYSA-N | | SMILES | C(O)(=O)C(OC)C |
| | 2-methoxypropionic acid Usage And Synthesis |
| Synthesis | A methanolic solution of 25% sodium methanolate (16 mL) was slowly added dropwise to a stirring solution of 2-bromopropionic acid (19.6 mmol) in methanol (5 mL) under nitrogen protection. The reaction mixture was heated and reacted at 50 °C overnight while continuously protected by the passage of nitrogen. Upon completion of the reaction, the reaction solution was concentrated under reduced pressure by rotary evaporator. The residue was adjusted to pH 1 with aqueous 1N hydrochloric acid, followed by three extractions with ethyl acetate (70 mL, 25 mL, 10 mL). The organic phases were combined, dried with anhydrous sodium sulfate, filtered and concentrated again under reduced pressure to afford 2-methoxypropionic acid as a colorless oil in the yield of 2.04 g (99% yield), which was pure enough to be used directly in the subsequent reaction without further purification. The product was characterized by 1H NMR (CD3OD): δ 3.67 (q, 1H), 3.33 (s, 3H), 1.33 ppm (d, 3H). | | References | [1] Patent: WO2006/96338, 2006, A1. Location in patent: Page/Page column 41
[2] Patent: WO2006/133006, 2006, A2. Location in patent: Page/Page column 46-47
[3] Australian Journal of Chemistry, 1980, vol. 33, # 4, p. 809 - 821
[4] Organic Letters, 2018, vol. 20, # 16, p. 4867 - 4870
[5] Patent: KR2015/128789, 2015, A. Location in patent: Paragraph 0896; 0898; 0899 |
| | 2-methoxypropionic acid Preparation Products And Raw materials |
|