|
|
| | Hexafluoropropylene Basic information |
| Product Name: | Hexafluoropropylene | | Synonyms: | 1,1,2,3,3,3-hexafluoro-1-propen;1-Propene, 1,1,2,3,3,3-hexafluoro-;1-Propene,1,1,2,3,3,3-hexafluoro-;CF3CF=CF2;Hexafluofoproene;hexafluoro-propen;Perfluoroprop-1-ene;HFP( hexafluoropropylene) | | CAS: | 116-15-4 | | MF: | C3F6 | | MW: | 150.02 | | EINECS: | 204-127-4 | | Product Categories: | API Intermediate;refrigerants;Fluoromonomer | | Mol File: | 116-15-4.mol | 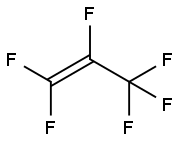 |
| | Hexafluoropropylene Chemical Properties |
| Melting point | −153 °C(lit.) | | Boiling point | −28 °C(lit.) | | density | 1,583 g/cm3 | | vapor pressure | 587.952kPa at 25℃ | | refractive index | 1.4000 | | form | gas | | Water Solubility | 82mg/L at 28℃ | | BRN | 1705671 | | Henry's Law Constant | 2.9×10-6 mol/(m3Pa) at 25℃, Wilhelm et al. (1977) | | Stability: | Stable. Incompatible with alkali metals, powdered metals. | | Major Application | PFAS testing | | InChI | InChI=1S/C3F6/c4-1(2(5)6)3(7,8)9 | | InChIKey | HCDGVLDPFQMKDK-UHFFFAOYSA-N | | SMILES | C(/F)(\F)=C(/F)\C(F)(F)F | | LogP | 1.95 at 20℃ | | CAS DataBase Reference | 116-15-4(CAS DataBase Reference) | | NIST Chemistry Reference | Propene, hexafluoro-(116-15-4) | | EPA Substance Registry System | Hexafluoropropene (116-15-4) | | ECETOC JACC REPORT | Hexafluoropropylene (116-15-4) |
| Hazard Codes | Xn,Xi | | Risk Statements | 20-37-68/20-48/20 | | Safety Statements | 41-36/37 | | RIDADR | UN 1858 (HEXAFLUOROPROPYLENE
(REFRIGERANT GAS R 1216)) | | WGK Germany | 1 | | RTECS | UD0350000 | | F | 4.5 | | Hazard Note | Irritant | | TSCA | TSCA listed | | HazardClass | 2.2 | | HS Code | 2903590030 | | Storage Class | 2A - Gases | | Hazard Classifications | Acute Tox. 4 Inhalation
Press. Gas Liquefied gas
STOT RE 2
STOT SE 2
STOT SE 3 | | Hazardous Substances Data | 116-15-4(Hazardous Substances Data) | | Toxicity | LC50 inhalation in mouse: 750ppm/4H |
| | Hexafluoropropylene Usage And Synthesis |
| Chemical Properties | Hexafluoropropylene (HFP) is a colourless, odourless, non-flammable gas that is only slightly soluble in water. It is used as chemical intermediate primarily in the synthesis of fluoropolymers, fluoro-elastomers and fluorinated materials (e.g. perfluoropolyether functional fluids, oils and greases). | | Uses | Hexafluoropropene is used in the synthesis of polyvinylidene fluoride-co-hexafluoropropylene, a polymer which is currently being used in various fields of the nano and electronic sector ranging from membranes to nanoparticles. | | Preparation | Hexafluoropropylene can be produced by pyrolysis of tetrafluoroethylene (TFE):
3 CF2=CF2 → 2 CF3CF=CF
This reaction is carried out in a closed continuous reactor at 600 to 900°C. The yield can be improved at low partial pressure, achieved either by operating under reduced total pressure (down to 0.1 bar) or by injecting an inert diluent such as steam or CO2.
Significant quantities of by-products, including the highly toxic perfluoroisobutylene (PFIB), are formed during the pyrolysis of TFE. Such by-products are either incinerated directly or are collected and transported for incineration at a remote facility.
Hexafluoropropylene can also be prepared from chlorodifluoromethane, or produced from various chlorofluorocarbons. | | Synthesis Reference(s) | Journal of the American Chemical Society, 73, p. 4054, 1951 DOI: 10.1021/ja01152a548 | | General Description | Hexafluoropropylene is an odorless, colorless gas. Hexafluoropropylene is noncombustible. Hexafluoropropylene can asphyxiate by the displacement of air. Exposure of the container to prolonged heat or fire can cause Hexafluoropropylene to rupture violently and rocket. | | Reactivity Profile | Halogenated aliphatic compounds, such as Hexafluoropropylene, are moderately or very reactive. Reactivity generally decreases with increased degree of substitution of halogen for hydrogen atoms. Materials in this group may be incompatible with strong oxidizing and reducing agents. Also, they may be incompatible with many amines, nitrides, azo/diazo compounds, alkali metals, and epoxides. Above a minimum oxygen pressure, the reaction of oxygen difluoride and hexafluoropropene to yield the Hexafluoropropylene oxide becomes explosive, Chem. Abs., 1987, 107, 175302. The reaction of hexafluoropropene with grignard reagent (subst. phenylmagnesium bromides) led to explosion, Fluorine Chem., 1981, 18, 25. | | Health Hazard | Vapors may cause dizziness or asphyxiation without warning. Vapors from liquefied gas are initially heavier than air and spread along ground. Contact with gas or liquefied gas may cause burns, severe injury and/or frostbite. Fire may produce irritating, corrosive and/or toxic gases. | | Fire Hazard | Some may burn but none ignite readily. Containers may explode when heated. Ruptured cylinders may rocket. | | Flammability and Explosibility | Non flammable |
| | Hexafluoropropylene Preparation Products And Raw materials |
| Raw materials | Tetrafluoroethylene | | Preparation Products | Perfluoroethylene propylene copolymer-->1,1,1,2,3,3,3-Heptafluoropropane-->(3,3,3-TRIFLUOROPROPYL)TRIMETHOXYSILANE-->Heptafluoroisopropyl iodide-->BIS(HEPTAFLUOROISOPROPYL)KETONE-->1,1,2,3,3,3-HEXAFLUOROPROPYL 2,2,2-TRIFLUOROETHYL ETHER-->HEPTAFLUORO-1-METHOXYPROPANE-->1H,1H,2'H-PERFLUORODIPROPYL ETHER-->1,1,2,3,3,3-HEXAFLUOROPROPYL METHYL ETHER-->PERFLUORO(2-METHYLPROPANOYL)FLUORIDE-->PERFLUOROPROPANE-->1,2-DIBROMOHEXAFLUOROPROPANE |
|