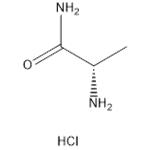
- ALA-NH2.HCL
-
- $0.00 / 1kg
-
2026-05-14
- CAS:33208-99-0
- Min. Order: 1kg
- Purity: 98+
- Supply Ability: 1T
|
| | L-Alaninamide hydrochloride Basic information |
| | L-Alaninamide hydrochloride Chemical Properties |
| Melting point | 212-217°C | | alpha | 11 º (c=1, MeOH) | | storage temp. | Inert atmosphere,Room Temperature | | solubility | DMSO (Slightly), Methanol (Slightly), Water | | form | Solid | | color | White to Off-White | | Optical Rotation | [α]25/D +11.0°, c = 1 in methanol | | Major Application | peptide synthesis | | InChI | InChI=1S/C3H8N2O.ClH/c1-2(4)3(5)6;/h2H,4H2,1H3,(H2,5,6);1H/t2-;/m0./s1 | | InChIKey | FIAINKIUSZGVGX-DKWTVANSSA-N | | SMILES | [C@H](N)(C)C(=O)N.Cl | | CAS DataBase Reference | 33208-99-0(CAS DataBase Reference) |
| Hazard Codes | C | | Safety Statements | 24/25 | | WGK Germany | 3 | | HS Code | 29241990 | | Storage Class | 11 - Combustible Solids |
| | L-Alaninamide hydrochloride Usage And Synthesis |
| Chemical Properties | white to light yellow crystal powder | | Uses | L-Alaninamide hydrochloride is used for preparation of novel amide derivatives of steroidal[3,2-c]pyrazole compounds with glucocorticoid activity. | | reaction suitability | reaction type: solution phase peptide synthesis | | Synthesis | The general procedure for the synthesis of L-alaninamide hydrochloride from alanine methyl ester hydrochloride is as follows: 500 g of methanol and 100 g of L-alanine were added to a 1000 ml three-necked flask, followed by the slow dropwise addition of 90 ml of thionyl chloride (the reaction temperature was controlled to be below 35 °C). After the dropwise addition, the three-necked flask was transferred to a water bath, heated to reflux temperature (temperature not exceeding 35 °C) and ensured that the generated SO2 was completely removed.Subsequently, the aminolysis reaction was carried out by passing 0.56 kg of ammonia into the reaction system under ice-bath conditions (temperature controlled below 20 °C), after which the reaction mixture was sealed and allowed to stand for 20 h in a room temperature water bath. The temperature of the water bath was raised to 55 °C to remove the remaining ammonia gas, which was finally concentrated to 300 ml and ensured that the ammonia gas was completely removed. Excess ammonium chloride was removed by filtration and the filter cake was washed with 100 ml of methanol, the washings were combined with the mother liquor and transferred to a 1000 ml beaker. The acidification process was completed by slowly adding 34.5 ml of hydrochloric acid at 10% concentration dropwise and adjusting the pH to 1.55. The reaction mixture was sealed and placed in a refrigerator for 12 h. After removal, 400 ml of acetone was added to induce crystallization. The crystals were collected by filtration, washed with an appropriate amount of solvent and subsequently dried at 45 °C for 12 h. 85 g of white crystals were obtained (85% yield). The rotativity of the product was [a] = +11.4°. | | References | [1] Journal fur Praktische Chemie - Chemiker - Zeitung, 1996, vol. 338, # 3, p. 251 - 256
[2] Patent: CN108069867, 2018, A. Location in patent: Paragraph 0015-0017 |
| | L-Alaninamide hydrochloride Preparation Products And Raw materials |
|