|
|
| | Pyrimidinetetramine sulfate Basic information |
| Product Name: | Pyrimidinetetramine sulfate | | Synonyms: | pyrimidinetetrayltetraamine sulphate;PYRIMIDINE-2,4,5,6-TETRAMINE SULPHATE;2,4,5,6-TETRAAMINIOPYRIMIDINE SULFATE;2,4,5,6-TETRAAMINO-PYRIMIDINE SULFATE SALT;2,4,5,6-Tetraaminopyrimidine sulfate hydrate,98%;2,4,5,6-Tetraaminopyrimidine sulfate;6-TetraaMinopyriMidine sulfate;PyriMidine-2,4,5,6-tetraaMine sulfate | | CAS: | 5392-28-9 | | MF: | C4H10N6O4S | | MW: | 238.22 | | EINECS: | 226-393-0 | | Product Categories: | Building Blocks;C4 to C5;Chemical Synthesis;Heterocyclic Building Blocks;Heterocycle-Pyrimidine series;Aromatics;Heterocycles;Intermediates;Building Blocks;Heterocyclic Building Blocks;FINE Chemical & INTERMEDIATES;Pyrimidines;Amines;(intermediate of methotrexate);bc0001 | | Mol File: | 5392-28-9.mol | 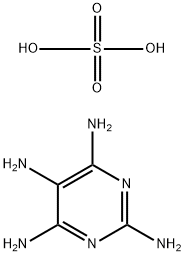 |
| | Pyrimidinetetramine sulfate Chemical Properties |
| Melting point | >300 °C(lit.) | | storage temp. | Sealed in dry,Room Temperature | | solubility | DMSO (Slightly, Heated), Water (Very Slightly, Heated) | | form | Crystalline Powder | | color | Ochre to brown | | Water Solubility | slightly soluble | | BRN | 3785189 | | Cosmetics Ingredients Functions | HAIR DYEING | | InChI | InChI=1S/C4H8N6.H2O4S/c5-1-2(6)9-4(8)10-3(1)7;1-5(2,3)4/h5H2,(H6,6,7,8,9,10);(H2,1,2,3,4) | | InChIKey | MQEFDQWUCTUJCP-UHFFFAOYSA-N | | SMILES | NC1=C(N=C(N)N=C1N)N.S(O)(O)(=O)=O | | CAS DataBase Reference | 5392-28-9(CAS DataBase Reference) | | EPA Substance Registry System | Pyrimidinetetramine, sulfate (1:1) (5392-28-9) |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 22-24/25-37/39-26-36 | | WGK Germany | 3 | | RTECS | UV9738000 | | TSCA | TSCA listed | | HS Code | 29335990 | | Storage Class | 11 - Combustible Solids |
| | Pyrimidinetetramine sulfate Usage And Synthesis |
| Chemical Properties | Light yellow crystal powder | | Uses | 2,4,5,6-Tetraaminopyrimidine Sulfate is an intermediate for the synthesis of the antitumor drugs methotrexate and fludarabine. | | Synthesis | Pyrimidinetetramine sulfate is synthesized from malononitrile and guanidine nitrate through a three-step reaction involving cyclization, nitrosation, and reduction. This process uses zinc powder instead of the expensive reducing agent Raney nickel, resulting in lower costs, simpler operation, and greater safety and environmental friendliness. | | Purification Methods | Purify the salt by recrystallisation from H2O, 2N H2SO4 (20 parts, 67% recovery) or 0.1N H2SO4 (40 parts, 62% recovery), and dried in air. [UV: Konrad & Pfleiderer Chem Ber 103 722 1970, Malletta et al. J Am Chem Soc 69 1814 1947, Cavalieri et al. J Am Chem Soc 70 3875 1948, Beilstein 25 H 423, 25 III/IV 3106.] | | References | [1] SAUMYA SHUKLA. Toxic potential assessment of hair dye developer 2,4,5,6-tetraaminopyrimidine sulfate exposed under ambient UVB radiation.[J]. Toxicology and Industrial Health, 2024: 1-8. DOI:10.1177/07482337231209352.
[2] https://ec.europa.eu/health/ph_risk/committees/04_sccp/docs/sccp_o_152.pdf |
| | Pyrimidinetetramine sulfate Preparation Products And Raw materials |
| Raw materials | 2,4,6-Triaminopyrimidine-->Ethyl cyanoacetate-->Urea-->4-Pyrimidinol, 2,6-diamino- (9CI)-->2,4,5-TRIAMINO-6-PYRIMIDINOL DIHYDROCHLORIDE | | Preparation Products | 6-(Bromomethyl)-2,4-pteridinediamine hydrobromide-->N10-(TRIFLUOROACETYL)PTEROIC ACID-->Pteridine (6CI,7CI,8CI,9CI)-->2,4-DIAMINO-6-(HYDROXYMETHYL)PTERIDINE-->2,4,5,6-TETRAAMINOPYRIMIDINE-->Methotrexate-->Fludarabine |
|