| Company Name: |
Shanghai EFE Biological Technology Co., Ltd.
|
| Tel: |
021-65675885 18964387627 |
| Email: |
info@efebio.com |
| Products Intro: |
Product Name:13,14-dihydro-15-keto Prostaglandin E2
CAS:363-23-5
Purity:95% Package:1mg;5mg;10mg
|
| Company Name: |
Shanghai Hongye Biotechnology Co. Ltd
|
| Tel: |
400-9205774 |
| Email: |
sales@glpbio.cn |
| Products Intro: |
Product Name:13,14-dihydro-15-keto Prostaglandin E2
CAS:363-23-5
Purity:>98% Package:1mg;10mg;50mg;100mg;
|
| Company Name: |
ChemeGen(Shanghai) Biotechnology Co.,Ltd.
|
| Tel: |
18818260767 |
| Email: |
sales@chemegen.com |
| Products Intro: |
Product Name:13,14-dihydro-15-keto Prostaglandin E2
CAS:363-23-5
Purity:98% Package:10 mg;50 mg;100 mg;500 mg;1 g;5 g;10 g
|
| Company Name: |
Hubei Kele Fine Chemical Co., Ltd
|
| Tel: |
027-59101668 19945030958 |
| Email: |
2223443625@qq.com |
| Products Intro: |
CAS:363-23-5
Purity:98% HPLC Package:1g;100g;1kg
|
|
| | 13,14-DIHYDRO-15-KETO PROSTAGLANDIN E2 Basic information |
| Product Name: | 13,14-DIHYDRO-15-KETO PROSTAGLANDIN E2 | | Synonyms: | 9,15-DIOXO-11ALPHA-HYDROXY-PROST-5Z-EN-1-OIC ACID;15-keto-13,14-dihydroprostaglandine2;13,14-dihydro-15-oxo-prostaglandin E2;CUJMXIQZWPZMNQ-XYYGWQPLSA-N;13,14-dihydro-15-keto Prostaglandin E2 MaxSpecStandard;13,14-DIHYDRO-15-KETO PROSTAGLANDIN E2;(E)-7-[(1R,2R,3R)-3-hydroxy-5-oxo-2-(3-oxooctyl)cyclopentyl]hept-5-enoic acid;13,14-Dihydro-15-keto-PGE2 | | CAS: | 363-23-5 | | MF: | C20H32O5 | | MW: | 352.47 | | EINECS: | | | Product Categories: | | | Mol File: | 363-23-5.mol | 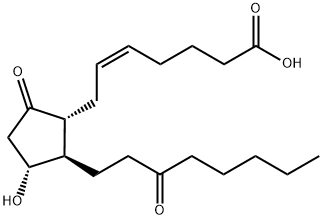 |
| | 13,14-DIHYDRO-15-KETO PROSTAGLANDIN E2 Chemical Properties |
| Boiling point | 538.1±45.0 °C(Predicted) | | density | 1.086±0.06 g/cm3(Predicted) | | storage temp. | −20°C | | solubility | DMF: >100 mg/ml
DMSO: >100 mg/ml
Ethanol: >100 mg/mlPBS (pH 7.2): >5 mg/ml | | form | Liquid | | pka | 4.75±0.10(Predicted) | | color | Colorless to light yellow |
| | 13,14-DIHYDRO-15-KETO PROSTAGLANDIN E2 Usage And Synthesis |
| Description | 13,14-dihydro-15-keto Prostaglandin E2 (13,14-dihydro-15-keto PGE2) is a metabolite of PGE2 and the primary PGE2 metabolite in plasma. It is formed from PGE2 via a 15-keto PGE2 intermediate by 15-oxo-PG Δ13 reductase. Unlike PGE2, 13,14-dihydro-15-keto PGE2 does not bind effectively to the PGE2 receptors EP2 and EP4 expressed in CHO cells (Kis = 12 and 57 μM, respectively) or induce adenylate cyclase activity in the same cells (EC50s = >18 and >38 μM, respectively). Levels of 13,14-dihydro-15-keto PGE2 are increased in the plasma of women in the third trimester of pregnancy and in women during and immediately after labor and delivery. Levels of 13,14-dihydro-15-keto PGE2 levels are decreased in tumor tissue compared to adjacent non-cancerous tissue isolated from patients with non-small cell lung cancer (NSCLC). | | Uses | 13,14-Dihydro-15-ketoPGE2 is a primary metabolite of PGE2 in plasma. | | Definition | ChEBI: The 13,14-dihydro derivative of 15-oxo-prostaglandin E2. | | References | [1] P. HUSSLEIN H. S. Concentration of 13,14-dihydro-15-keto-prostaglandin E2 in the maternal peripheral plasma during labour of spontaneous onset[J]. Bjog-An International Journal of Obstetrics and Gynaecology, 1984, 91 3: 228-231. DOI: 10.1111/j.1471-0528.1984.tb04757.x
[2] DUNCAN HUGHES. NAD+-dependent 15-hydroxyprostaglandin dehydrogenase regulates levels of bioactive lipids in non-small cell lung cancer.[J]. Cancer prevention research (Philadelphia, Pa.), 2008, 1 4: 241-249. DOI: 10.1158/1940-6207.capr-08-0055
[3] JOVANA MARIC PHD , PHD Gerd G M Nerea Ferreirós PhD, PHD Danielle F M, et al. Cytokine-induced endogenous production of prostaglandin D2 is essential for human group 2 innate lymphoid cell activation[J]. Journal of Allergy and Clinical Immunology, 2019, 143 6: Pages 2202-2214.e5. DOI: 10.1016/j.jaci.2018.10.069
[4] M. HAMBERG B S. On the Metabolism of Prostaglandins E1 and E2 in Man[J]. Journal of Biological Chemistry, 1971, 246 1: 6713-6721. DOI: 10.1016/s0021-9258(19)45905-x
[5] N. NISHIGAKI A I M Negishi. Two Gs-coupled prostaglandin E receptor subtypes, EP2 and EP4, differ in desensitization and sensitivity to the metabolic inactivation of the agonist.[J]. Molecular Pharmacology, 1996, 146 1: 1031-1037. DOI: 10.1254/fpj.108.supplement_65
[6] DAVID MERIWETHER. Apolipoprotein A-I mimetics mitigate intestinal inflammation in COX2-dependent inflammatory bowel disease model.[J]. Journal of Clinical Investigation, 2019, 129 9: 3670-3685. DOI: 10.1172/jci123700 |
| | 13,14-DIHYDRO-15-KETO PROSTAGLANDIN E2 Preparation Products And Raw materials |
|