- 5-Iodouracil
-

- $0.00 / 5mg
-
2026-03-12
- CAS:696-07-1
- Min. Order:
- Purity:
- Supply Ability: 10g
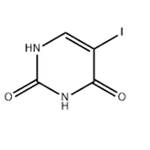
- 5-Iodouracil
-
- $0.00 / 1KG
-
2025-04-04
- CAS:696-07-1
- Min. Order: 1KG
- Purity: 98%
- Supply Ability: 1ton
- 5-Iodouracil
-

- $100.00 / 1Kg/Bag
-
2022-02-25
- CAS:696-07-1
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 100KG
|
| | 5-Iodouracil Basic information |
| | 5-Iodouracil Chemical Properties |
| Melting point | 274-276 °C (dec.) (lit.) | | density | 2.2076 (estimate) | | storage temp. | 2-8°C | | solubility | Very faint turbidity in NH3aq. Soluble in 1M NaOH. | | pka | 7.02±0.10(Predicted) | | form | Fluffy Powder | | color | White to light yellow | | Water Solubility | SOLUBLE IN COLD WATER | | Sensitive | Light Sensitive | | BRN | 4891 | | InChI | InChI=1S/C4H3IN2O2/c5-2-1-6-4(9)7-3(2)8/h1H,(H2,6,7,8,9) | | InChIKey | KSNXJLQDQOIRIP-UHFFFAOYSA-N | | SMILES | C1(=O)NC=C(I)C(=O)N1 | | CAS DataBase Reference | 696-07-1(CAS DataBase Reference) | | NIST Chemistry Reference | 5-Iodouracil(696-07-1) | | EPA Substance Registry System | 2,4(1H,3H)-Pyrimidinedione, 5-iodo- (696-07-1) |
| Hazard Codes | T,Xi | | Risk Statements | 46-20/21/22-36/37/38 | | Safety Statements | 53-22-26-36/37/39-45 | | RIDADR | 2811 | | WGK Germany | 3 | | RTECS | YR0525000 | | Hazard Note | Irritant/Carcinogenic/Light Sensitive | | TSCA | TSCA listed | | HazardClass | 6.1 | | PackingGroup | III | | HS Code | 29335990 | | Storage Class | 13 - Non Combustible Solids | | Hazard Classifications | Acute Tox. 4 Dermal
Acute Tox. 4 Inhalation
Acute Tox. 4 Oral
Eye Irrit. 2
Skin Irrit. 2
STOT SE 3 |
| | 5-Iodouracil Usage And Synthesis |
| Chemical Properties | white to light yellow fluffy powder | | Uses | 5-Iodouracil is a halogenated pyrimidine that can be used in nucleoprotein photo-crosslinking via RNA substitution. 5-Iodouracil is used in thymidine phosphorylase targeted imaging and therapy. Studies show that DNA N-glycosylase MED1 exhibited higher preference for 5-Iodouracil and halogenated bases over non-halogenated ones. | | Definition | ChEBI: An organoiodine compound consisting of uracil having an iodo substituent at the 5-position. | | Synthesis | The general procedure for the synthesis of 5-iodouracil (5-IUra) from pyrimidine-2,4(1H,3H)-dione (Ura) is as follows:
Example 1 Synthesis of 5-halogenated uracil (5-X-Ura)
Preparation of 5-iodouracil (5-IUra) and 5-bromouracil (5-BrUra) from Ura:
1. Prepare a solution of 1,3,4,6-tetrachloro-3a,6a-diphenylglycuronium in chloroform (concentration 0.5 mg/mL). All reagents were available from Sigma Chemical Co. (St. Louis, MO) unless otherwise noted.
2. Dry 0.5 mL (containing 250 μg, 578 μmol) of the above solution using a stream of nitrogen.
3. 300 μL of Ura (2610 μmol, 8.7 M, dissolved in 0.25 M potassium phosphate buffer, pH 7.5) was added.
4. 100 μL NaI or NaBr (670 μmol, 6.7 M aqueous solution) was subsequently added to the reaction system.
5. The reaction mixture was heated at 60 °C for 15 min.
6. Upon completion of the reaction, the yield was 79% for 5-IUra and 56% for 5-BrUra, depending on the halide used.
7. At the end of the reaction, the reaction mixture was diluted with water and the products were analyzed by reversed-phase high performance liquid chromatography (HPLC).
8. HPLC mobile phase conditions: 20-50 mM acetic acid and 6-16% acetonitrile (ACN).
Note: This method is suitable for the synthesis of materials on a small scale and also for the therapeutic isotope labeling of Ura by replacing non-radioactive halides with radioisotopes such as [82Br] or [125I]. | | References | [1] Tetrahedron Letters, 2002, vol. 43, # 8, p. 1381 - 1386
[2] Synlett, 2005, # 8, p. 1263 - 1266
[3] Helvetica Chimica Acta, 2015, vol. 98, # 7, p. 953 - 960
[4] Synthesis, 2004, # 11, p. 1869 - 1873
[5] Synthesis, 1995, # 8, p. 926 - 928 |
| | 5-Iodouracil Preparation Products And Raw materials |
|