
5-FLUOROINDOLE-3-CARBOXYLIC ACID synthesis
- Product Name:5-FLUOROINDOLE-3-CARBOXYLIC ACID
- CAS Number:23077-43-2
- Molecular formula:C9H6FNO2
- Molecular Weight:179.15

1260793-83-6

23077-43-2
General procedure for the synthesis of 5-fluoro-1H-indole-3-carboxylic acid from 2,2,2-trifluoro-1-(5-fluoro-1H-indol-3-yl)ethanone: 2,2,2-trifluoro-1-[5-fluoro-1-(pyrimidin-2-yl)-1H-indol-3-yl]ethan-1-one (1.6 g) and 5 N aqueous sodium hydroxide were added to the reaction flask. The reaction mixture was stirred under heated reflux conditions for 1.5 hours. After completion of the reaction, the reaction solution was cooled to room temperature and extracted by adding water and ethyl acetate. After separating the organic and aqueous layers, the aqueous layer was made acidic with concentrated hydrochloric acid. The acidic aqueous layer was again extracted with ethyl acetate, the organic layers were combined and washed with saturated saline. The organic layer was dried with anhydrous sodium sulfate and the solvent was removed by distillation under reduced pressure to give 785 mg of 5-fluoro-1H-indole-3-carboxylic acid (88% yield).

1260793-83-6
15 suppliers
inquiry

23077-43-2
114 suppliers
$27.00/100mg
Yield:23077-43-2 785 mg
Reaction Conditions:
with sodium hydroxide in water; for 1.5 h;Reflux;
Steps:
113-2 (113-2) Synthesis of 5-Fluoro-lH-indole-3-carboxylic acid(Compound 113-2)
2,2,2 trifluoro-1-[5-fluoro-l- (pyrimidin-2-yl)-1H-indol-3-yl] ethan-1-one(1.6 g)Was added 5 N sodium hydroxide aqueous solution (20 mL)And the mixture was stirred under heating reflux for 1.5 hours.After cooling the reaction solution to room temperature, water and ethyl acetate were added,And extracted with ethyl acetate. The obtained aqueous layer was made acidic with concentrated hydrochloric acid,The mixture was extracted with ethyl acetate and washed with saturated brine. The organic layer was dried over sodium sulfate and the solvent was distilled off under reduced pressure to give 5-fluoro-lH-indole-3-carboxylic acid785 mg (yield 88%) was obtained.
References:
JP2017/171619,2017,A Location in patent:Paragraph 0168; 0170
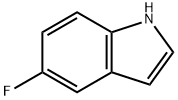
399-52-0
381 suppliers
$10.00/1g

124-38-9
134 suppliers
$214.00/14L

23077-43-2
114 suppliers
$27.00/100mg

399-52-0
381 suppliers
$10.00/1g

407-25-0
473 suppliers
$9.00/1g

23077-43-2
114 suppliers
$27.00/100mg

399-52-0
381 suppliers
$10.00/1g

23077-43-2
114 suppliers
$27.00/100mg
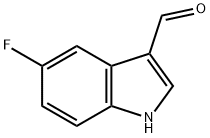
2338-71-8
169 suppliers
$15.00/250mg

23077-43-2
114 suppliers
$27.00/100mg