
Ethyl-2-(diphenylaMino)pyriMidine-5-carboxylate synthesis
- Product Name:Ethyl-2-(diphenylaMino)pyriMidine-5-carboxylate
- CAS Number:1316216-05-3
- Molecular formula:C19H17N3O2
- Molecular Weight:319.36

591-50-4
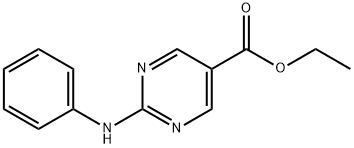
864172-93-0

1316216-05-3
The general procedure for the synthesis of ethyl 2-(diphenylamino)pyrimidine-5-carboxylate from iodobenzene and compound (CAS: 864172-93-0) was as follows: compound 2 (6.2 g, 25 mmol), iodobenzene (6.12 g, 30 mmol), CuI (955 mg, 5.0 mmol), Cs2CO3 (16.3 g, 50 mmol) The mixture in TEOS (200 mL) was degassed and purged with nitrogen. The resulting mixture was stirred and reacted at 140 °C for 14 hours. After completion of the reaction, it was cooled to room temperature and the reaction mixture was diluted with EtOAc (200 mL) and 95% EtOH (200 mL) and filtered through a pre-prepared column of NH4F-H2O silica gel [50 g, prepared by adding NH4F (100 g) to water (1500 mL) followed by silica gel (500 g, 100-200 mesh)]. The mixture was allowed to stand for 2 h at room temperature before being filtered and the solid was collected and washed with EtOAc. The filtrate was concentrated to dryness and the residue was purified by silica gel column chromatography (petroleum ether/EtOAc = 10:1) to give a yellow solid product (3 g, 38% yield).

591-50-4
500 suppliers
$10.00/1g

864172-93-0
3 suppliers
inquiry

1316216-05-3
28 suppliers
$10.00/250mg
Yield:1316216-05-3 69.9%
Reaction Conditions:
with copper (I) iodide;Cs2CO3 in N,N-dimethyl-formamide at 140; for 12 h;Inert atmosphere;
Steps:
1-2 Synthesis of Intermediate 3:
In a 500ml three-necked flask, add compound 2 (17.00g, 70mmol),Iodobenzene (17.11g, 84mmol), cesium carbonate (45.54g, 140mmol),Cuprous iodide (6.65g, 35mmol), DMF 200ml, under nitrogen protection at 140 for 12h.Cool to room temperature, add 200 ml of ethyl acetate to dilute, filter to remove copper salt, add 200 ml of water to the filtrate, stir at room temperature for 20 min, separate the liquid, and extract the aqueous phase twice with 100 ml of ethyl acetate.Combine the organic phases, add 200ml of EDTA saturated solution, stir for 30min, and separate the organic layer.Wash with 100ml of saturated saline, concentrate to dryness under reduced pressureObtained brown oil, recrystallized by adding 50ml of ethanol,15.6g light yellow solid was obtained,The yield was 69.90%.
References:
CN110885316,2020,A Location in patent:Paragraph 0095; 0102-0104; 0127-0130

89793-12-4
234 suppliers
$6.00/100mg

1316216-05-3
28 suppliers
$10.00/250mg
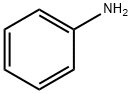
62-53-3
688 suppliers
$10.00/1g

1316216-05-3
28 suppliers
$10.00/250mg

95928-49-7
92 suppliers
$45.00/50mg

1316216-05-3
28 suppliers
$10.00/250mg