Trypsin
- CAS No.
- 9002-07-7
- Chemical Name:
- Trypsin
- Synonyms
- 500mg;TRYPSIN-EDTA;EC 3.4.21.1;TRYPSIN PORCINE;RecoMbinant Trypsin;trypsin from porcine pancreas;TRL3;Lys-C;C00298;u-4858
- CBNumber:
- CB0673677
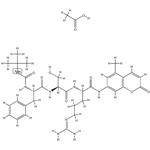
- Molecular Formula:
- C35H47N7O10
- Molecular Weight:
- 725.78858
- MDL Number:
- MFCD01323069
- MOL File:
- 9002-07-7.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| Trypsin from bovine pancreas powder, ≥7,500?BAEE units/mg solid | T9201 | 100MG | $131 |
| Trypsin from bovine pancreas powder, ≥7,500?BAEE units/mg solid | T9201 | 500MG | $215 |
| Trypsin from bovine pancreas powder, ≥7,500?BAEE units/mg solid | T9201 | 1G | $332 |
| Trypsin from bovine pancreas powder, ≥7,500?BAEE units/mg solid | T9201 | 5G | $922 |
| Trypsin from bovine pancreas powder, ≥7,500?BAEE units/mg solid | T9201 | 10G | $1470 |
| More product size | |||
| Melting point | 115°C |
|---|---|
| bulk density | 200kg/m3 |
| Density | 1.37[at 20℃] |
| vapor pressure | 0Pa at 25℃ |
| storage temp. | -20°C |
| solubility | Reconstitute in aqueous buffer |
| pka | pK1:6.25 (25°C,μ=0.1) |
| form | lyophilized powder |
| color | White powder |
| Odor | Odorless |
| PH | 7.70-8.30 |
| biological source | Porcine pancreas |
| Water Solubility | Soluble in water (10 mg/ml), phosphate buffers (10 mg/ml), and balanced salt solutions (1 mg/ml). |
| Merck | 13,9865 |
| Specific Activity | 90-110% (compared to standard) |
| Stability | Stable. Incompatible with strong oxidizing agents. |
| Major Application | diagnostic assay manufacturing |
| Cosmetics Ingredients Functions |
SKIN CONDITIONING HAIR CONDITIONING |
| LogP | -1.3 at 20℃ |
| FDA 21 CFR | 184.1914 |
| CAS DataBase Reference | 9002-07-7 |
| Substances Added to Food (formerly EAFUS) | TRYPSIN FROM ANIMAL TISSUE |
| EWG's Food Scores | 4 |
| FDA UNII | V6GZ69J3FW |
| ATC code | B06AA07,D03BA01,M09AB52 |
| EPA Substance Registry System | Trypsin (9002-07-7) |
| UNSPSC Code | 41116107 |
| NACRES | NA.78 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H334 | |||||||||
| Precautionary statements | P261-P284-P304+P340+P312-P501 | |||||||||
| target organs | Respiratory system | |||||||||
| PPE | dust mask type N95 (US), Eyeshields, Faceshields, Gloves | |||||||||
| Hazard Codes | Xn,B | |||||||||
| Risk Statements | 36/37/38-42-42/43 | |||||||||
| Safety Statements | 22-24-26-36/37-45-23 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | GC3050000 | |||||||||
| F | 1-3-10 | |||||||||
| TSCA | TSCA listed | |||||||||
| HS Code | 35079090 | |||||||||
| Storage Class | 11 - Combustible Solids | |||||||||
| Hazard Classifications | Eye Irrit. 2 Resp. Sens. 1 Skin Irrit. 2 STOT SE 3 |
|||||||||
| Hazardous Substances Data | 9002-07-7(Hazardous Substances Data) | |||||||||
| NFPA 704 |
|
Trypsin price More Price(94)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | T9201 | Trypsin from bovine pancreas powder, ≥7,500?BAEE units/mg solid | 9002-07-7 | 100MG | $131 | 2026-04-30 | Buy |
| Sigma-Aldrich | T9201 | Trypsin from bovine pancreas powder, ≥7,500?BAEE units/mg solid | 9002-07-7 | 500MG | $215 | 2026-04-30 | Buy |
| Sigma-Aldrich | T9201 | Trypsin from bovine pancreas powder, ≥7,500?BAEE units/mg solid | 9002-07-7 | 1G | $332 | 2026-04-30 | Buy |
| Sigma-Aldrich | T9201 | Trypsin from bovine pancreas powder, ≥7,500?BAEE units/mg solid | 9002-07-7 | 5G | $922 | 2026-04-30 | Buy |
| Sigma-Aldrich | T9201 | Trypsin from bovine pancreas powder, ≥7,500?BAEE units/mg solid | 9002-07-7 | 10G | $1470 | 2026-04-30 | Buy |
Trypsin Chemical Properties,Uses,Production
Description
Trypsin is a serine protease in the digestive system of human and animals. The main function of this enzyme is to hydrolyze proteins into smaller peptides or even amino acids. Trypsin and other digestive proteases such as chymotrypsin are responsible for the digestion of food protein in the small intestine. This proteolytic function of trypsin has been widely used in the protein chemistry, proteomics, and nutrition research. This function is influenced by the sources of enzyme, and environmental factors such as pH, temperature, and the presence of trypsin inhibitors in the enzymatic reaction medium.
Trypsin is used in the food processing to improve the functional properties such as solubility, emulsification, foaming and gelling properties of food proteins, to improve the digestibility of vegetable and seed proteins. It is used to reduce the concentration of allergens in some foods and to produce protein hydrolysates and bioactive peptides that are used in infant formulas and for people with special health problems such as hypertension. In food science research, trypsin is used for the food protein sequencing, in-vitro determination of food protein digestibility. In combination with bromelain and rutin, trypsin is used for osteoarthritis. Trypsin is used to remove necrotic tissue and debris during wound and ulcer cleaning. Trypsin supplements may be used to remove dead tissue cells that remain after trauma, infection or surgical procedures, allowing new skin or tissue cells to grow.
References
[1] http://www.cytospring.com/pages/TrypsinEDTA.pdf
[2] Jianmei Yu, Mohamed Ahmedna (2012) Functions/applications of trypsin in food processing and food science research, 75-95
[3] http://www.webmd.com/vitamins-supplements/ingredientmono-879-trypsin.aspx?activeingredientid=879&activeingredientname=trypsin
Chemical Properties
White or almost white, crystalline or amorphous powder, hygroscopic if amorphous.
Uses
Proteolytic enzyme.
Uses
Trypsin is a digestive enzyme found in the digestive system. The enzyme catalyzes the hydrolysis of peptide bonds breaking down proteins into smaller peptides.
Uses
Trypsin-EDTA Solution 10X has been used to release adherent cells from tissue culture plates for passaging.
Definition
trypsin: An enzyme that digests proteins(see protease). It is secreted inan inactive form (trypsinogen) by thepancreas into the duodenum. There,trypsinogen is acted on by an enzyme(enterokinase) produced in theduodenum to yield trypsin. The activeenzyme plays an important rolein the digestion of proteins in the anteriorportion of the small intestine.It also activates other proteases inthe pancreatic juice.
brand name
Parenzyme;Trypsillin.
General Description
Trypsin is applicable for tissue disaggregation, due to its effective action and tolerance towards different cell type and serum-induced neutralization.
Biochem/physiol Actions
Trypsin cleaves peptides on the C-terminal side of lysine and arginine residues. The rate of hydrolysis of this reaction is slowed if an acidic residue is on either side of the cleavage site and hydrolysis is stopped if a proline residue is on the carboxyl side of the cleavage site. The optimal pH for trypsin activity is 7-9. Trypsin can also act to cleave ester and amide linkages of synthetic derivatives of amino acids. EDTA is added to trypsin solutions as a chelating agent that neutralizes calcium and magnesium ions that obscure the peptide bonds on which trypsin acts. Removing these ions increases the enzymatic activity. Serine protease inhibitors, including DFP, TLCK, APMSF, AEBSEF, and aprotinin, amongst others, will inhibit Trypsin.
Description
Recombinant Porcine Trypsin is expressed in E.coli and purified by standard chromatography techniques.
Source
Ecoli
Applications
Trypsin digestion: the suggested ratio is 1:50 to 1:1000 (w/w).
Background
Trypsin (EC3.4.21.4) is part of the serine protease family. Trypsin cleaves lysine and arginine at the C-terminal side of the peptide. The hydrolysis rate is slower if an acidic residue is on either sides of the cleavage site and no cleavage occurs if a proline residue is on the carboxyl side of the cleavage site. Trypsin optimum pH is pH-7 to 9. Trypsin will also hydrolyze ester and amide linkages of synthetic derivatives of amino acids such as: benzoyl L-arginine ethyl ester (BAEE), p-toluenesulfonyl- L-arginine methyl ester (TAME), tosyl-L-arginine methyl ester, N-α-benzoyl-L-arginine p-nitroanilide (BAPNA), L-lysyl-p-nitroanilide, and benzoyl-L-tyrosine ethyl ester (BTEE). Serine protease inhibitors that inhibit recombinant trypsin include TLCK (N-p-tosyl-L-lysine chloromethyl ketone), PMSF (phenylmethanesulfonyl fluoride), benzamidine, soybean trypsin inhibitor, and ovomucoid.
Trypsin Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Aladdin Scientific | tp@aladdinsci.com | United States | 57505 | 58 | |
| Hebei Chuanghai Biotechnology Co., Ltd | +8615350571055 | Sibel@chuanghaibio.com | China | 8738 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +8617392709771 | 1097@dideu.com | China | 3996 | 58 |
| Hebei Chuanghai Biotechnology Co,.LTD | +86-86-13131129325 +8613131129325 | sales1@chuanghaibio.com | China | 5235 | 58 |
| airuikechemical co., ltd. | +86-18353166132 | sales02@airuikechemical.com | China | 983 | 58 |
| Hebei Zhuanglai Chemical Trading Co.,Ltd | +8613343047651 | admin@zlchemi.com | China | 3692 | 58 |
| Hebei Longbang Technology Co., LTD | +86-18633929156 | admin@hblongbang.com | China | 972 | 58 |
| HebeiShuoshengImportandExportco.,Ltd | +86-18532138899 | L18532138899@163.com | China | 939 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21590 | 55 |
| Shanxi Naipu Import and Export Co.,Ltd | +86-13734021967 +8613734021967 | kaia@neputrading.com | China | 1001 | 58 |
Related articles
- Trypsin in colorectal cancer
- Trypsin is overexpressed in cancer and contributes to invasiveness. In colorectal cancer, trypsin overexpression correlates wi....
- Jan 24,2024
- Trypsin: Preparation and Application Studies
- Trypsin is an important pancreatic serine protease in mammals. The main function of this enzyme is to hydrolyze proteins into ....
- Apr 18,2023
View Lastest Price from Trypsin manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2026-05-04 | Trypsin
9002-07-7
|
0.99 | RongNa Biotechnology Co.,Ltd | ||||
 |
2026-04-30 | Trypsin
9002-07-7
|
US $0.00 / KG | 1KG | 2500ups u/mg | 500kg/month | WUHAN FORTUNA CHEMICAL CO., LTD | |
 |
2026-04-20 | Trypsin
9002-07-7
|
US $39.00-89.00 / mg | 10g | TargetMol Chemicals Inc. |





