9-Bromoanthracene
- CAS No.
- 1564-64-3
- Chemical Name:
- 9-Bromoanthracene
- Synonyms
- 9-BROMOANTHRENE;9-BROMOANTHRACENE;9-Anthryl bromide;MS-BROMOANTHRACENE;10-Bromoanthracene;9-Bromo anthrecene;TIMTEC-BB SBB007978;RARECHEM AQ BD AN02;9-Anthracyl bromide;Anthracene, 9-bromo-
- CBNumber:
- CB5319891
- Molecular Formula:
- C14H9Br
- Molecular Weight:
- 257.13
- MDL Number:
- MFCD00001243
- MOL File:
- 1564-64-3.mol
- MSDS File:
- SDS
| Product description | Number | Pack Size | Price |
| 9-Bromoanthracene 94% | B56609 | 5g | $87.3 |
| 9-Bromoanthracene 94% | B56609 | 25g | $379 |
| 9-Bromoanthracene >95.0%(GC) | B0872 | 5g | $17 |
| 9-Bromoanthracene >95.0%(GC) | B0872 | 25g | $54 |
| 9-Bromoanthracene >99.0%(GC)(HPLC) | B2871 | 5g | $30 |
| More product size | |||
| Melting point | 97-100 °C (lit.) |
|---|---|
| Boiling point | 303.85°C (rough estimate) |
| Density | 1.4251 (rough estimate) |
| refractive index | 1.6404 (estimate) |
| storage temp. | Sealed in dry,Room Temperature |
| solubility | Chloroform (Slightly), DMSO (Slightly), Methanol (Slightly) |
| form | Powder |
| color | Yellow |
| Water Solubility | Insoluble in water. |
| BRN | 1869747 |
| InChI | InChI=1S/C14H9Br/c15-14-12-7-3-1-5-10(12)9-11-6-2-4-8-13(11)14/h1-9H |
| InChIKey | ZIRVQSRSPDUEOJ-UHFFFAOYSA-N |
| SMILES | C1=C2C(C=C3C(=C2Br)C=CC=C3)=CC=C1 |
| CAS DataBase Reference | 1564-64-3(CAS DataBase Reference) |
| FDA UNII | 4KA5657H57 |
| NIST Chemistry Reference | 9-Bromoanthracene(1564-64-3) |
| UNSPSC Code | 12352100 |
| NACRES | NA.22 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H341 | |||||||||
| Precautionary statements | P201-P202-P280-P308+P313-P405-P501 | |||||||||
| PPE | Eyeshields, Gloves, type N95 (US) | |||||||||
| Hazard Codes | Xi | |||||||||
| Safety Statements | 22-24/25 | |||||||||
| WGK Germany | 3 | |||||||||
| Hazard Note | Irritant | |||||||||
| HS Code | 29049090 | |||||||||
| Storage Class | 11 - Combustible Solids | |||||||||
| NFPA 704 |
|
9-Bromoanthracene price More Price(40)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | B56609 | 9-Bromoanthracene 94% | 1564-64-3 | 5g | $87.3 | 2026-03-19 | Buy |
| Sigma-Aldrich | B56609 | 9-Bromoanthracene 94% | 1564-64-3 | 25g | $379 | 2026-03-19 | Buy |
| TCI Chemical | B0872 | 9-Bromoanthracene >95.0%(GC) | 1564-64-3 | 5g | $17 | 2026-03-19 | Buy |
| TCI Chemical | B0872 | 9-Bromoanthracene >95.0%(GC) | 1564-64-3 | 25g | $54 | 2026-03-19 | Buy |
| TCI Chemical | B2871 | 9-Bromoanthracene >99.0%(GC)(HPLC) | 1564-64-3 | 5g | $30 | 2026-03-19 | Buy |
9-Bromoanthracene Chemical Properties,Uses,Production
Description
9-bromoanthracene is a kind of bromine-derived anthracene. It is known to be able to reversibly photodimerize in a head-to tail fashion upon irradiation by long-wavelength ultraviolet light. The photodimers of 9-bromoanthracene are suitable to be used as alkyl halide initiators in the atom transfer radical polymerization (ATRP) reactions. It is also used as the intermediate for the preparation of the 9-substitued form of the polycyclic aromatic hydrocarbon (PAH) anthracene.
Chemical Properties
white to light yellow crystal powder. boiling point 190°C (0.16kPa, sublimation), relative density 1.409. soluble in acetic acid; carbon disulfide.
Uses
9-Bromoanthracene acts as an intermediate in the preparation of 9-substituted derivative of the polycyclic aromatic hydrocarbon (PAH) anthracene.
Preparation
Anthracene (5 g, 28.05 mmol) was dissolved in CHCl3. Then N-bromosuccinimide (NBS,4.99 g, 28.05 mmol) was added in batches away from light, and the reaction solution was continuously stirred for 12 h. The resulting mixture was stirred for another 30 min with appropriate water, and extracted with CH2Cl2. The CH2Cl2 solution was dried over anhydrous MgSO4. After removing CH2Cl2 solvent, the residue was recrystallized from anhydrous ethanol to give 4.78 g (66.3 %) of a green-yellow needle solid 9-Bromoanthracene. 1H NMR (500 MHz, CDCl3) δ 8.55 (d, J = 8.9 Hz, 2H), 8.48 (s, 1H), 8.03 (d, J = 8.4 Hz, 2H), 7.67 – 7.60 (m, 2H), 7.56 – 7.51 (m, 2H). EI-MS (m/z): Calculated for C14H9Br: 257.13. Found [M+ ]: 255.96.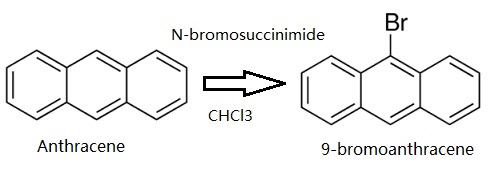
Synthesis of 9-bromoanthracene
Application
The Sonogashira reaction of 9-Bromoanthracene and ethynyltrimethylsilane gave not only the expected anticipated 9-(trimethylsilylethynyl)anthracene and 2-(trimethylsilyl)aceanthrylene and (E)-4-(9-anthracenyl)-1,3-bis(trimethylsilyl)-but-3-en-1-yne were also obtained in moderate yields. The mechanistic principle may be that the coupling of bromo(anthracenyl)bis(triphenylphosphine)palladium(II) can occur either directly or after coordination and migratory insertion of the free alkyne.
Synthesis Reference(s)
The Journal of Organic Chemistry, 57, p. 2740, 1992 DOI: 10.1021/jo00035a038
Synthesis

120-12-7

1564-64-3
General procedure for the synthesis of 9-bromoanthracene from anthracene: A mixture of anthracene (356 mg, 2 mmol), potassium bromide (143 mg, 1.2 mmol), acetic acid (9 mL), water (1 mL), and dichloromethane (5 mL) was added sequentially to a three-necked flask equipped with a condenser. A thermometer was inserted into the reaction system and the flask was transferred to a thermostatically heated magnetic stirring water bath. ZnAl-BrO3-LDHs (1.8 g, 1.8 mmol) was added slowly in batches over 15 min. The reaction temperature was controlled to be 40 °C. The reaction progress was monitored by thin layer chromatography (TLC). After 5 h of reaction, the reaction solution was washed with sodium sulfite solution and then extracted with dichloromethane (3 × 10 mL). The organic phases were combined and two scoops of column chromatography silica gel (200-300 mesh) were added to the dichloromethane phase and the organic solvent was removed by evaporation. Purification by column chromatography (eluent was petroleum ether: ethyl acetate = 15:1) under vacuum gave 468 mg of the target product 9-bromoanthracene as a pale yellow solid in 91% yield.
Purification Methods
Crystallise 9-bromoanthracene from MeOH or EtOH followed by sublimation in vacuo. [Masnori et al. J Am Chem Soc 108 126 1986, Beilstein 5 IV 2295.]
Properties and Applications
9-Bromoanthracene could be used to synthesise 6-bromoaceanthrylene-1,2-dione by Friedel-Crafts acylation reaction with oxalyl chloride. The pure 9-Bromoanthracene’s energy level is -5.710 eV. A 1:1 co-crystal of Picric acid and 9-Bromoanthracene was prepared by a solvent evaporation method. The PIC: BRA co-crystal shows significantly reduced impact sensitivity relative to pure Picric acid. The primary non-covalent interaction occurs between the electron-poor π-system of Picric acid and the electron-rich π-system of 9-Bromoanthracene. The distance between the Picric acid centroid and the 9-Bromoanthracene centroid was measured to be 3.492 Å, a typical value in materials which exhibit aromatic face-to-face πinteractions (<4.00 Å). An interaction is also indicated between aromatic hydrogen on the 9-Bromoanthracene ring and nitro group oxygen on a parallel Picric acid ring (C3-H3…O6). The donor-acceptor π ... π interactions dominate the formation of the Picric Acid co-crystal. The π electron cloud of the 9-Bromoanthracene ring is concentrated towards the electron-poor region at the centre of the Picric acid moiety, and the two rings combine. The π system of picric acid is known to deform due to the electron-withdrawing effect of nitro groups and an electron vacancy formed at the centre of the ring. This is partially filled by π electrons of the 9-Bromoanthracene ring. In other words, a charge transformation complex is formed[4].
References
[1]Cohen, Nicole A., et al. Macromolecular Chemistry and Physics 210.3 ‐4 (2009): 263-268.
[2]Xu, Xiaoming, Wenzhe Lu, and Richard B. Cole. Analytical chemistry 68.23 (1996): 4244-4253.
[3]Dang, Hung, and Miguel A. Garcia-Garibay. Journal of the American Chemical Society 123.2 (2001): 355-356.
[4] Se Hun Kim. “Highly efficient deep-blue emitting organic light emitting diode based on the multifunctional fluorescent molecule comprising covalently bonded carbazole and anthracene moieties.” Journal of Materials Chemistry B 13 1 (2011): 9139–9148.
9-Bromoanthracene Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Springchem New Material Technology Co.,Limited | +86-021-62885108 +8613917661608 | info@spring-chem.com | China | 2068 | 57 |
| Shandong Xingshun New Material Co., Ltd. | +86-0519-86461196 | gxx@xingshengtech.com | China | 552 | 58 |
| Zhengzhou HQ Material CO.,LTD. | +86-371-55919009 +8615225108189 | sale1@hqmat.com | China | 1001 | 58 |
| Zhengzhou Lingzhiyue Technology Co., Ltd | +86-0371-55074660 +86-13523716970 | Lingzhiyue@aliyun.com | China | 3145 | 58 |
| SHANGHAI KEAN TECHNOLOGY CO., LTD. | +8613817748580 | cooperation@kean-chem.com | China | 40066 | 58 |
| Hebei Xinsheng New Material Technology Co., LTD. | +86-16632316109 | xinshengkeji@xsmaterial.com | China | 1085 | 58 |
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 | info@fdachem.com | China | 20124 | 58 |
| Capot Chemical Co.,Ltd. | +86-(0)57185586718; +8613336195806 | sales@capot.com | China | 29640 | 60 |
| Shanghai Daken Advanced Materials Co.,Ltd | +86-2158073036 | info@dakenam.com | China | 13815 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21592 | 55 |
Related articles
- 9-Bromoanthracene: Synthesis, Impurity Analysis and Thermochemical Behavior Studies
- 9-Bromoanthracene acts as an intermediate in the preparation of 9-substituted derivative of the polycyclic aromatic hydrocarbo....
- Apr 24,2025
View Lastest Price from 9-Bromoanthracene manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2025-09-25 | 9-Bromoanthracene
1564-64-3
|
US $100.00-1.00 / KG | 1KG | 99% | g-kg-tons, free sample is available | Henan Fengda Chemical Co., Ltd | |
 |
2025-07-02 | Anthracene, 9-bromo-
1564-64-3
|
US $0.00 / KG | 1KG | 98.00% | 150KG /month | Shanghai Daken Advanced Materials Co.,Ltd | |
 |
2025-06-04 | 9-Bromoanthracene
1564-64-3
|
US $0.00-0.00 / kg | 1kg | 98%+ | 10000kgs per Month | Shanghai Qyubiotech Co., Ltd. |
-

- 9-Bromoanthracene
1564-64-3
- US $100.00-1.00 / KG
- 99%
- Henan Fengda Chemical Co., Ltd
-

- Anthracene, 9-bromo-
1564-64-3
- US $0.00 / KG
- 98.00%
- Shanghai Daken Advanced Materials Co.,Ltd
-

- 9-Bromoanthracene
1564-64-3
- US $0.00-0.00 / kg
- 98%+
- Shanghai Qyubiotech Co., Ltd.
1564-64-3(9-Bromoanthracene)Related Search:
1of4