|
|
| | 2-Cyano-N,N-diethylacetamide Basic information |
| Product Name: | 2-Cyano-N,N-diethylacetamide | | Synonyms: | N,N-DIETHYLCYANOACETAMIDE;2-Cyano-N,N-diethylacetamide;2-Cyano-N,N-diethylacetaMid;N,N-diethyl caynoacetaMide;N,N-Diethyl-2-cyanoacetoamide;2-Cyano-N,N-diethylacetamide;2-Cyano-N,N-diethylacetamide >;Acetamide, 2-cyano-N,N-diethyl- | | CAS: | 26391-06-0 | | MF: | C7H12N2O | | MW: | 140.18 | | EINECS: | | | Product Categories: | Aliphatics;Miscellaneous Reagents;pharmaceutical intermediates | | Mol File: | 26391-06-0.mol | 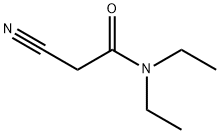 |
| | 2-Cyano-N,N-diethylacetamide Chemical Properties |
| Melting point | 121 °C | | Boiling point | 114°C/4mmHg(lit.) | | density | 1.013 g/mL at 25 °C | | refractive index | n20/D1.466 | | Fp | >110℃ | | storage temp. | 2-8°C | | solubility | Chloroform, Ethyl Acetate, Tetrahydrofuran, Methanol | | pka | 4.53±0.10(Predicted) | | form | Oil | | color | Light Yellow to Light Brown | | InChI | InChI=1S/C7H12N2O/c1-3-9(4-2)7(10)5-6-8/h3-5H2,1-2H3 | | InChIKey | RYSHIRFTLKZVIH-UHFFFAOYSA-N | | SMILES | C(N(CC)CC)(=O)CC#N | | CAS DataBase Reference | 26391-06-0(CAS DataBase Reference) |
| WGK Germany | 3 | | HazardClass | IRRITANT | | HS Code | 2926907090 | | Storage Class | 10 - Combustible liquids |
| | 2-Cyano-N,N-diethylacetamide Usage And Synthesis |
| Chemical Properties | Pale Brown Oil | | Uses | N,N-Diethyl-2-cyanoacetoamide can be used:
- To prepare N,N-diethyl 2-cyano-2-fluoro-2-phenylthioacetoamide, which is further used in the synthesis of various cyanofluoroamides.
- In the synthesis of entacapone, a catechol-O-methyltransferase inhibitor by reacting with 3,4-dihydroxy-5-nitrobenzaldehyde.
- As a building block in the synthesis of fluorophores, applicable as endoplasmic reticulum probes.
| | Definition | ChEBI: N,N-diethylcyanoacetamide is an aliphatic nitrile and a monocarboxylic acid amide. | | Synthesis | (i) Preparation of N1,N1-diethyl-2-cyanoacetamide: 30 L of anhydrous tetrahydrofuran (THF) was added to the reactor under nitrogen protection, followed by diethylamine (1.272 kg, 17.39 mol). A 33% hexane solution of lithium n-hexyl was loaded into a dropping funnel. The reactor was cooled to below -30°C. The hexyl lithium solution was slowly added dropwise to the reactor, keeping the temperature of the reaction mixture below -30°C during the dropwise addition. After the dropwise addition was completed, the reaction mixture was continued to be stirred at -30 °C for 1.5 hours. Meanwhile, ethyl cyanoacetate (0.655 kg, 5.79 mol) was dissolved in THF (2 L) and transferred to another dropping funnel. The ethyl cyanoacetate solution was slowly added dropwise to the reaction mixture at a temperature well below -30°C. After completion of the dropwise addition, the reaction mixture was gradually warmed to room temperature and stirring was continued for 30 minutes. Subsequently, ethanol (0.66 L) was slowly added dropwise to the reaction mixture and stirred overnight at room temperature. The solvent was removed by distillation under reduced pressure and the residue was treated with 10% hydrochloric acid (8 L) and extracted twice with dichloromethane (DCM, 2 x 7 L). The DCM layers were combined and washed with water (2 × 5 L). The DCM solution was dried and evaporated to dryness to afford the brown oily product N1,N1-diethyl-2-cyanoacetamide (755.29 g, 97% yield), which could be used in the subsequent steps without further purification.1H-NMR (200 MHz, CDCl3): δ 1.2 ppm (t, 6H, J = 7Hz), 3.4 ppm (q, 4H, J = 7Hz), 3.55ppm (s, 2H). | | References | [1] Patent: US2008/103191, 2008, A1. Location in patent: Page/Page column 17
[2] Australian Journal of Chemistry, 1976, vol. 29, p. 1039 - 1050
[3] Synthetic Communications, 1990, vol. 20, # 20, p. 3235 - 3243
[4] Journal of the Korean Chemical Society, 2012, vol. 56, # 4, p. 459 - 463 |
| | 2-Cyano-N,N-diethylacetamide Preparation Products And Raw materials |
|