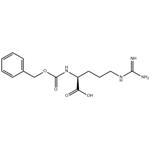
- Cbz-Arg-OH
-

- $0.00/ kg
-
2026-03-26
- CAS:1234-35-1
- Min. Order: 1kg
- Purity: 98%
- Supply Ability: 1T+
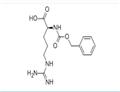
- Nalpha-Cbz-L-Arginine
-
- $0.00 / 1KG
-
2025-12-24
- CAS:1234-35-1
- Min. Order: 1KG
- Purity: 99.0%
- Supply Ability: 10000KG
- Nalpha-Cbz-L-Arginine
-
- $1.00 / 1KG
-
2019-07-06
- CAS:1234-35-1
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 20kg
|
| | Nalpha-Cbz-L-Arginine Basic information |
| | Nalpha-Cbz-L-Arginine Chemical Properties |
| Melting point | 171-174 °C (dec.)(lit.) | | alpha | -11 º (c=0.5, 0.5N HCl 24 ºC) | | Boiling point | 448.73°C (rough estimate) | | density | 1.1765 (rough estimate) | | refractive index | -10 ° (C=5.5, 0.2mol/L HCl) | | storage temp. | 2-8°C | | solubility | DMSO, Water | | pka | 3.90±0.21(Predicted) | | form | Solid | | color | White | | Optical Rotation | [α]20/D 9±1°, c = 5% in 1 M HCl | | BRN | 2169267 | | Major Application | peptide synthesis | | InChI | 1S/C14H20N4O4/c15-13(16)17-8-4-7-11(12(19)20)18-14(21)22-9-10-5-2-1-3-6-10/h1-3,5-6,11H,4,7-9H2,(H,18,21)(H,19,20)(H4,15,16,17)/t11-/m0/s1 | | InChIKey | SJSSFUMSAFMFNM-NSHDSACASA-N | | SMILES | NC(=N)NCCC[C@H](NC(=O)OCc1ccccc1)C(O)=O | | CAS DataBase Reference | 1234-35-1(CAS DataBase Reference) | | EPA Substance Registry System | L-Arginine, N2-[(phenylmethoxy)carbonyl]- (1234-35-1) |
| | Nalpha-Cbz-L-Arginine Usage And Synthesis |
| Chemical Properties | white powder | | Uses | Z-L-Arginine is used in the synthesis of renin inhibitors containing phosphorous acid derivatives at the scissile bond. Also used in the synthesis of a peptide deformylase inhibitor BB-3497. | | reaction suitability | reaction type: solution phase peptide synthesis | | Synthesis | The general procedure for the synthesis of Cbz-arginine from cyanamide and (S)-5-amino-2-(benzyloxycarbonyloxyamino)pentanoic acid was as follows: to a solution of water (2.5 mL) containing p-methoxyaniline (1b, 61.6 mg, 0.50 μmol) and cyanamide (25.2 mg, 0.60 μmol) was added scandium trifluoromethanesulfonate (Sc(OTf)3, at room temperature. 24.6 mg, 50 nmol). The reaction mixture was stirred at 100 °C for 2 days. After completion of the reaction, the reaction mixture was washed with chloroform (3 x 10 mL) extraction. The aqueous layer was separated and concentrated under vacuum. The residue was purified by filtration through a silica gel pad (eluent: chloroform-methanol, 20:1, v/v). | | References | [1] Synlett, 2014, vol. 25, # 9, p. 1302 - 1306 |
| | Nalpha-Cbz-L-Arginine Preparation Products And Raw materials |
|