- L(+)-Leucinol
-

- $0.00 / 1KG
-
2026-04-29
- CAS:7533-40-6
- Min. Order: 1KG
- Purity: 98.0%
- Supply Ability: 50kg/month
- H-L-Leu-ol
-
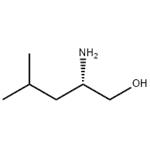
- $0.00/ kg
-
2026-04-29
- CAS:7533-40-6
- Min. Order: 1kg
- Purity: 98%
- Supply Ability: 1T+
- L(+)-Leucinol
-

- $30.00 / 10mg
-
2026-04-22
- CAS:7533-40-6
- Min. Order:
- Purity: 98.00%
- Supply Ability: 10g
|
| | L(+)-Leucinol Basic information |
| | L(+)-Leucinol Chemical Properties |
| Melting point | 56-58 °C | | alpha | 3 º (589nm, c=9, ethanol) | | Boiling point | 208-210 °C(lit.) | | density | 0.917 g/mL at 25 °C(lit.) | | refractive index | n20/D 1.4511(lit.) | | Fp | 195 °F | | storage temp. | Keep in dark place,Inert atmosphere,2-8°C | | solubility | Soluble in Chloroform,Dichloromethane,Ethyl Acetate,DMSO,Acetone,etc. | | form | Liquid | | pka | 12.88±0.10(Predicted) | | color | Clear colorless to slightly yellow light | | Specific Gravity | 0.917 | | Optical Rotation | [α]20/D +4°, c = 9 in ethanol | | Sensitive | Air Sensitive | | BRN | 1719240 | | Major Application | peptide synthesis | | InChI | 1S/C6H15NO/c1-5(2)3-6(7)4-8/h5-6,8H,3-4,7H2,1-2H3/t6-/m0/s1 | | InChIKey | VPSSPAXIFBTOHY-LURJTMIESA-N | | SMILES | CC(C)C[C@H](N)CO | | CAS DataBase Reference | 7533-40-6(CAS DataBase Reference) | | NIST Chemistry Reference | 1-Pentanol, 2-amino-4-methyl-, (S)-(7533-40-6) |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 26-36-36/37/39-27 | | WGK Germany | 3 | | F | 2-10 | | HS Code | 29221990 | | Storage Class | 10 - Combustible liquids | | Hazard Classifications | Eye Irrit. 2
Skin Irrit. 2
STOT SE 3 |
| | L(+)-Leucinol Usage And Synthesis |
| Chemical Properties | clear colorless to slightly yellow | | Uses | Starting material for the synthesis of aminopeptidase N and phospholipase A2 inhibitors. | | reaction suitability | reaction type: solution phase peptide synthesis | | Synthesis | The general procedure for the synthesis of (S)-2-amino-4-methylpentan-1-ol from L-leucine methyl ester hydrochloride was as follows: to a mixture of EtOH (1.8 L) containing L-leucine methyl ester hydrochloride (254 g, 1.4 mol), NaHCO3 (118 g, 1.4 mol, 1.0 eq.), and water (1.8 L) at 5°C, batchwise addition of NaBH4 (159 g, 4.2 mol, 3.0 eq.) at a rate commensurate with the reaction temperature, maintaining the reaction temperature at no more than 15°C (ca. 70 min.) After the addition of NaBH4 was complete, the ice bath was removed, and the reaction mixture was heated to reflux temperature and held overnight. Upon completion of the reaction, the mixture was cooled to room temperature. The resulting slurry was filtered with the aid of an ice bath and the solids were washed with EtOH (750 mL). The filtrates were combined and concentrated under reduced pressure to about 950 mL. the residue was diluted with EtOAc (2.5 L) and subsequently extracted with 1N NaOH solution (2 x 1 L). The aqueous layer was back-extracted with EtOAc (2 x 750mL). The organic phases were combined, dried over MgSO4 and concentrated under reduced pressure to afford (S)-1-(hydroxymethyl)-3-methylbutylamine as a light yellow oil (112 g, 65% yield). The product was characterized by 1H NMR (CDCl3): δ 0.88-0.93 (m, 6H), 1.17 (t, J = 7.7 Hz, 2H), 1.68-1.80 (m, 2H), 1.82 (br s, 2H), 2.86-2.91 (m, 1H), 3.22 (dd, J = 10.7,8.1 Hz, 1H), 3.56 ( dd, J = 10.3,3.6 Hz, 1H). | | References | [1] Patent: US6353006, 2002, B1. Location in patent: Page column 31 |
| | L(+)-Leucinol Preparation Products And Raw materials |
|