|
|
| | tert-Butyl 4-(2-ethoxy-2-oxoethyl)piperidine-1-carboxylate Basic information |
| Product Name: | tert-Butyl 4-(2-ethoxy-2-oxoethyl)piperidine-1-carboxylate | | Synonyms: | N-BOC-4-ETHYL PIPERIDINECARBOXYLATE;tert-butyl 4-(2-ethoxy-2-oxoethyl)piperidine-1-carboxylate(SALTDATA: FREE);Ethyl N-Boc-4-piperidineacetate;Ethyl 1-Boc-4-piperidine ...;Ethyl 1-Boc-4-piperidine acetate;1-[(tert-butoxy)carbonyl]-4-ethylpiperidine-2-carboxylate;tert-Butyl 4-(2-ethoxy-2-oxoethyl)piperidin-1-carboxylate;4-Piperidineacetic acid, 1-[(1,1-diMethylethoxy)carbonyl]-, ethyl ester | | CAS: | 135716-09-5 | | MF: | C14H25NO4 | | MW: | 271.35 | | EINECS: | | | Product Categories: | | | Mol File: | 135716-09-5.mol | 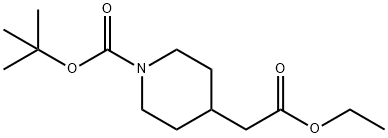 |
| | tert-Butyl 4-(2-ethoxy-2-oxoethyl)piperidine-1-carboxylate Chemical Properties |
| Melting point | 19.68° C (Predicted) | | Boiling point | 339.2±15.0 °C(Predicted) | | density | 1.048±0.06 g/cm3(Predicted) | | refractive index | n20D 1.47 (Predicted) | | storage temp. | Sealed in dry,Room Temperature | | pka | -1+-.0.40(Predicted) | | form | solid | | Appearance | Colorless to light yellow Liquid | | InChI | 1S/C14H25NO4/c1-5-18-12(16)10-11-6-8-15(9-7-11)13(17)19-14(2,3)4/h11H,5-10H2,1-4H3 | | InChIKey | PQEXLIRUMIRSAL-UHFFFAOYSA-N | | SMILES | CCOC(=O)CC1CCN(CC1)C(=O)OC(C)(C)C |
| Hazard Codes | Xn | | Risk Statements | 22 | | HazardClass | IRRITANT | | Storage Class | 11 - Combustible Solids | | Hazard Classifications | Acute Tox. 4 Oral |
| | tert-Butyl 4-(2-ethoxy-2-oxoethyl)piperidine-1-carboxylate Usage And Synthesis |
| Uses |
tert-Butyl 4-(2-ethoxy-2-oxoethyl)piperidine-1-carboxylate is used as a building block for the synthesis of organic compounds and drugs.
| | Synthesis | Tert-butyl 4-(2-ethoxy-2-oxoethylidene)piperidine-1-carboxylate (22 g, 81.8 mmol) was added to wet 10% Pd/C (2.2 g) and ethanol (160 mL), and the mixture was degassed and backfilled three times with hydrogen. The reaction mixture was stirred at room temperature for 2 h under the protection of a hydrogen balloon. Upon completion of the reaction, the catalyst was removed by filtration and the filtrate was concentrated to afford tert-butyl 4-(2-ethoxy-2-oxoethyl)piperidine-1-carboxylate (22.5 g, 100% yield). The m/z (M-100) was 172.1 as detected by LRMS, which was consistent with the theoretical value of 172.1. | | References | [1] Heterocycles, 2001, vol. 54, # 2, p. 747 - 755
[2] Patent: WO2018/68297, 2018, A1. Location in patent: Page/Page column 135; 136
[3] Patent: WO2018/152329, 2018, A1. Location in patent: Page/Page column 48
[4] Journal of the Chemical Society, Perkin Transactions 1: Organic and Bio-Organic Chemistry (1972-1999), 1995, # 6, p. 641 - 644
[5] Chinese Chemical Letters, 2012, vol. 23, # 6, p. 707 - 710 |
| | tert-Butyl 4-(2-ethoxy-2-oxoethyl)piperidine-1-carboxylate Preparation Products And Raw materials |
|