|
|
| | Chloro(1,5-cyclooctadiene)iridium(I) dimer Basic information | | Reactions |
| Product Name: | Chloro(1,5-cyclooctadiene)iridium(I) dimer | | Synonyms: | DI-MU-CHLOROBIS[(ETA-CYCLOOCTA-1,5-DIENE)IRIDIUM (I)];DI-MU-CHLORO-BIS[(1,2,5,6-ETA)-1,5-CYCLOOCTADIENE]DIIRIDIUM;CHLORO(1,5-CYCLOOCTADIENE)IRIDATE (I) DIMER;CHLORO(1,5-CYCLOOCTADIENE)IRIDIUM(I) DIMER;IRIDIUM(I) CHLORIDE 1,5-CYCLOOCTADIENE COMPLEX DIMER;IRIDIUM I CYCLOOCTADIENE CHLORIDE;IRIDIUM CHLORO-1,5-CYCLOOCTADIENE;IRIDIUM COD CHLORIDE | | CAS: | 12112-67-3 | | MF: | C16H24Cl2Ir2 | | MW: | 671.7 | | EINECS: | 235-170-7 | | Product Categories: | Ir;organometallic complexes;Ir (Iridium) Compounds;Catalysts for Organic Synthesis;Classes of Metal Compounds;Homogeneous Catalysts;Metal Complexes;Synthetic Organic Chemistry;Transition Metal Compounds | | Mol File: | 12112-67-3.mol | 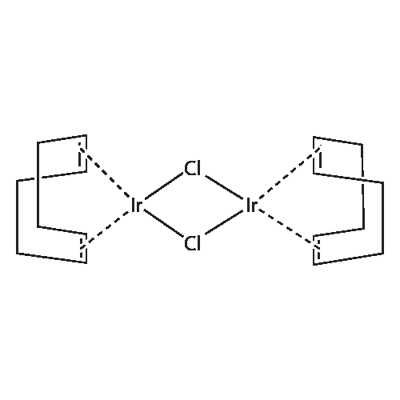 |
| | Chloro(1,5-cyclooctadiene)iridium(I) dimer Chemical Properties |
| Melting point | 205 °C (dec.)(lit.) | | storage temp. | Keep in dark place,Inert atmosphere,2-8°C | | solubility | Chloroform (Slightly), Methanol (Slightly) | | form | Powder | | color | red to orange | | Water Solubility | insoluble | | Hydrolytic Sensitivity | 7: reacts slowly with moisture/water | | InChI | InChI=1S/2C8H12.2ClH.2Ir/c2*1-2-4-6-8-7-5-3-1;;;;/h2*1-2,7-8H,3-6H2;2*1H;;/q;;;;2*+1/p-2/b2*2-1-,8-7-;;;; | | InChIKey | XHOSESNLNGITPM-XRGHXPOKSA-L | | SMILES | C1C=CCCC=CC1.C1C=CCCC=CC1.Cl[Ir].Cl[Ir] |c:1,5,9,13| |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 26-37/39 | | WGK Germany | 3 | | F | 10 | | TSCA | No | | HS Code | 28439000 | | Storage Class | 11 - Combustible Solids |
| | Chloro(1,5-cyclooctadiene)iridium(I) dimer Usage And Synthesis |
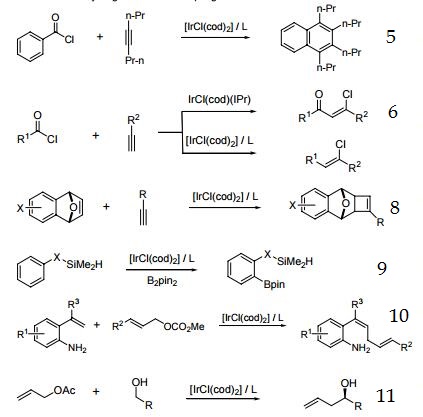
| Reactions | 1. Precursor to catalysts for the asymmetric hydrogenation of tri- and tetrasubstituted olefins.
2. Precursor to catalyst for enantioselective reduction of imines.
3. Precursor to catalyst for allylic alkylation.
4. Precursor to catalyst for allylic amination and etherification.
5. Precursor to catalyst for the reaction of aroyl chlorides with internal alkynes to produce substituted naphthalenes and anthracenes.
6. Ir-catalyzed addition of acid chlorides to terminal alkynes.
7. Intramolecular hydroamination of unactivated alkenes with secondary alkyl- and arylamines.
8. Enantioselective [2+2] cycloaddition.
9. Silyl-directed, Ir-catalyzed ortho-borylation of arenes.
10. Ir-catalyzed cross-coupling of styrene derivatives with allylic carbonates.
11. Transfer hydrogenative C-C coupling
| | Chemical Properties | red-orange solid | | Uses | Chloro(1,5-cyclooctadiene)iridium(I) dimer is widely used as a precursor to other iridium complexes, which finds application in homogeneous catalysis like carbonylation, hydrosilylation, hydrofomylation, asymmetric allylic substitutions, metathesis and chiral catalysis reactions. It is involved in the preparation of Crabtree's catalyst, which is used for hydrogenation and hydrogen-transfer reactions. | | reaction suitability | core: iridium
reaction type: C-H Activation
reagent type: catalyst | | Synthesis |
A solution of 6 mL of 1,5-cyclooctadiene in 35 mL of ethanol and 20 mL of water is added to 2.0 g of IrCl3.3H2O in a round-bottomed flask. The mixture is refluxed under nitrogen for 24 h when an orange-red product precipitates from the solution. The mixture is cooled to rt, and Di-μ-chlorobis(1,5-cyclooctadiene)diiridium(I) (Chloro(1,5-cyclooctadiene)iridium(I) dimer) is collected by filtration, washed with cold methanol, and dried in vacuo at 25 °C for 8 h.
| | References | [1] Patent: CN106220688, 2016, A. Location in patent: Paragraph 0049-0051 |
| | Chloro(1,5-cyclooctadiene)iridium(I) dimer Preparation Products And Raw materials |
| Raw materials | Tetrahydrofuran-->Isopropyl alcohol-->Hydrazinium hydroxide solution-->Hydroquinone-->1,5-Cyclooctadiene-->Iridium(III) chloride hydrate-->Iridium, aquatetrachloro--->[1E,5Z,pR]-1,5-Cyclooctadiene-->Benzaldehyde | | Preparation Products | [(S)-(-)-5,5'-Bis(diphenylphosphino)-4,4'-bi-1,3-benzodioxole][4-cyano-3-nitrobenzenecarboxylato][1,2,3-η-2-propenyl]iridiuM(III), Min. 98% |
|