|
|
| | 4-Bromo-2-methyl-benzaldehyde Basic information | | Uses |
| Product Name: | 4-Bromo-2-methyl-benzaldehyde | | Synonyms: | 4-Bromo-2-methylbenzaldehyde ,98%;5-Bromo-2-formyltoluene;5-Bromo-2-formyltoluene, 4-Bromo-o-tolualdehyde;Benzaldehyde,4-broMo-2-Methyl-;Methyl-4-bromobenzaldehyde;4-BROMO-2-METHYLBENZALDEHYDE >97%;4-Bromo-2-methyl-benzaldehyde, 97%min | | CAS: | 24078-12-4 | | MF: | C8H7BrO | | MW: | 199.04 | | EINECS: | | | Product Categories: | | | Mol File: | 24078-12-4.mol | 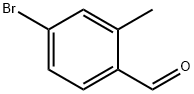 |
| | 4-Bromo-2-methyl-benzaldehyde Chemical Properties |
| Boiling point | 115-116 °C(Press: 8 Torr) | | density | 1.490±0.06 g/cm3(Predicted) | | storage temp. | Inert atmosphere,2-8°C | | form | Solid | | Appearance | White to yellow <25°C Solid,>25°C Liquid | | InChI | InChI=1S/C8H7BrO/c1-6-4-8(9)3-2-7(6)5-10/h2-5H,1H3 | | InChIKey | RCBPVESMGNZMSG-UHFFFAOYSA-N | | SMILES | C(=O)C1=CC=C(Br)C=C1C |
| | 4-Bromo-2-methyl-benzaldehyde Usage And Synthesis |
| Uses | 4-Bromo-2-methylbenzaldehyde has a wide range of chemical transformation properties and can be used as an intermediate in organic synthesis, for example, it can be used in the preparation of progesterone receptor modulators. | | Chemical Properties | solid | | Chemical Reactivity | 4-Bromo-2-methyl-benzaldehyde is an organic compound that can be synthesized by the transfer hydrogenation of 2-methylcyclohexanone. It is used as an additive in the production of polymers and plastics. Its catalytic properties make it a good candidate for use as a hydrogenation catalyst and in sustainable chemical synthesis. | | Synthesis | Procedure for the synthesis of 4-bromo-2-methylbenzaldehyde: Oxalyl chloride (249 mL, 497 mmol) was dissolved in dichloromethane (150 mL) under argon protection and cooled to -78 °C. A dichloromethane solution (75 mL) of dimethyl sulfoxide (DMSO, 42.4 mL, 597 mmol) was slowly added dropwise through a vent needle, taking care to control the rate of dropwise acceleration to avoid violent outgassing. After dropwise addition, the venting needle was removed and the reaction mixture continued to be stirred at -78 °C for 30 min. Subsequently, a dichloromethane solution (203 mL) of (4-bromo-2-methylphenyl)methanol (28A, 20.0 g, 99.0 mmol) was added and stirred for 30 min at -78 °C. Next, triethylamine (TEA, 166 mL, 1190 mmol) was slowly added dropwise and the reaction mixture was gradually warmed to room temperature and stirring was continued for 12 hours. After completion of the reaction, the organic layer was separated by diluting the reaction mixture with dichloromethane (30 mL) and water (20 mL). The aqueous layer was extracted with dichloromethane (3 x 50 mL), the organic phases were combined, washed sequentially with water and brine, dried over anhydrous magnesium sulfate, filtered and concentrated. Purification of the crude product by fast chromatography afforded the target compound 4-bromo-2-methylbenzaldehyde (28B, 15.4 g, 78 mmol, 78% yield) as a yellow oil.1H NMR (400 MHz, CDCl3) δ 10.22 (s, 1H), 7.66 (d, J = 8.28 Hz, 1H), 7.51 (dd, J = 8.28, 1.76 Hz, 1H). 1.76 Hz, 1H), 7.45 (s, 1H), 2.65 (s, 3H). | | References | [1] Patent: WO2014/78608, 2014, A1. Location in patent: Paragraph 00281
[2] Patent: WO2015/171757, 2015, A1. Location in patent: Page/Page column 55
[3] Journal of Medicinal Chemistry, 1984, vol. 27, # 11, p. 1516 - 1531
[4] Patent: WO2008/91681, 2008, A2. Location in patent: Page/Page column 225
[5] Patent: US2010/16298, 2010, A1. Location in patent: Page/Page column 191 |
| | 4-Bromo-2-methyl-benzaldehyde Preparation Products And Raw materials |
|