|
|
| | (S,S)-(+)-N,N'-BIS(3,5-DI-TERT-BUTYLSALICYLIDENE)-1,2-CYCLOHEXANEDIAMINO-COBALT(II) Basic information | | Reaction |
| Product Name: | (S,S)-(+)-N,N'-BIS(3,5-DI-TERT-BUTYLSALICYLIDENE)-1,2-CYCLOHEXANEDIAMINO-COBALT(II) | | Synonyms: | (1S,2S)-(+)-1,2-CYCLOHEXANEDIAMINO-N N'-BIS(3,5-DI-T-BUTYLSALICYLIDENE)COBALT(II);(1S,2S)-(+)-N,N'-BIS(3,5-DI-T-BUTYLSALICYDENE)-1,2-CYCLOHEXANEDIAMINOCOBALT(II);(S,S)-(+)-N,N'-BIS(3,5-DI-TERT-BUTYLSALICYLIDENE)-1,2-CYCLOHEXANEDIAMINO-COBALT(II);(1S,2S)-(+)-N,N-Bis(3,5-di-t-butylsalicylidene)-1,2-cyclohexanediaminocobalt(II);(S,S)-N,N'-BIS(3,5-DI-T-BUTYLSALICYLIDEN E) -1,2-CYCLOHEXANEDIAMINOCOBALT(II);(1S,2S)-(+)-1,2-Cyclohexanediamino-N,N'-bis(3,5-di-tert-butylsalicylidene)cobalt(II);(S,S)-(+)-N,N'-Bis(3,5-di-tert-butylsalicylidene)-1,2-cyclohexanediaminocobalt(II),98%;(S,S)-(+)-N,N'-Bis(3,5-di-tert-butylsalicylidene)-1,2-cyclohexane | | CAS: | 188264-84-8 | | MF: | C36H52CoN2O2 | | MW: | 603.76 | | EINECS: | | | Product Categories: | Co;Jacobsen Metal;fine chemicals, specialty chemicals, intermediates, electronic chemical, organic synthesis;Asymmetric Synthesis;Chiral Catalysts, Ligands, and Reagents;Kinetic ResolutionChiral Catalysts, Ligands, and Reagents;Privileged Ligands and Complexes;SALENs;chem001 | | Mol File: | 188264-84-8.mol |  |
| | (S,S)-(+)-N,N'-BIS(3,5-DI-TERT-BUTYLSALICYLIDENE)-1,2-CYCLOHEXANEDIAMINO-COBALT(II) Chemical Properties |
| Melting point | >350 °C(lit.) | | storage temp. | Keep in dark place,Inert atmosphere,Room temperature | | form | Powder | | color | red-brown | | InChIKey | ZFWPDJKMASHRPT-DKZUAMKGNA-L | | SMILES | C(C1C=C(C(C)(C)C)C=C2C=N3[C@@]4([H])CCCC[C@]4([H])N4=CC5=CC(C(C)(C)C)=CC(C(C)(C)C)=C5[O-][Co+2]43[O-]C=12)(C)(C)C |&1:12,18,r| |
| Hazard Codes | Xn | | Risk Statements | 20/21/22 | | Safety Statements | 36/37/39-36/37 | | WGK Germany | 3 | | Storage Class | 11 - Combustible Solids |
| | (S,S)-(+)-N,N'-BIS(3,5-DI-TERT-BUTYLSALICYLIDENE)-1,2-CYCLOHEXANEDIAMINO-COBALT(II) Usage And Synthesis |
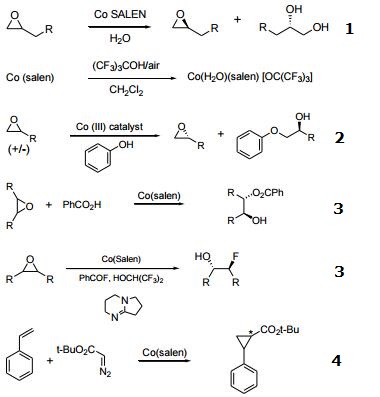
| Reaction |
- Catalyst used in the kinetic resolution of racemic, terminal epoxides yielding a chiral diol and the unreacted enantiomer of the epoxide.
- Precursor to a Co(III) catalyst for the kinetic resolution of terminal epoxides with alcohols.
- Desymmetrization of meso-epoxides with carboxylic acids and fluoride.
- Catalyst for asymmetric cyclopropanation of styrene.
- Catalyst for copolymerization of CO2 and epoxides.
- Enantioselective intramolecular openings of oxetanes.

| | Chemical Properties | red to red-brown powder | | Uses | (S,S)-(+)-N,N''-Bis(3,5-di-tert-butylsalicylidene)-1,2-cyclohexanediaminocobalt(ii) i sused as a catalyst for the hydrolytic kinetic resolution of terminal epoxides and the enantioselective ring opening of meso epoxides. | | Synthesis | The general procedure for the synthesis of (S,S)-N,N'-bis(3,5-di-tert-butylsalicylidene)-1,2-cyclohexanediamine cobalt(II) from cobalt acetate and 1,2-cyclohexylamine bis(3,5- butyl sulphate) is as follows: with reference to Example 1, (R,R)-N,N'-bis(3,5-di-tert-butylsalicylidene)-1,2-cyclohexanediamine (10.9 g. 20.0 mmol) was dissolved in dichloromethane (80 mL). The solution was slowly added to a methanol solution of cobalt tetrahydrate acetate (5.98 g, 24.0 mmol) (80 mL) and the reaction was stirred at room temperature for 15 min. A red solid gradually precipitated during the reaction. The reaction mixture was then continued to be stirred at 0 °C for 30 min to promote complete precipitation. The red solid was collected by filtration and the target product Co(L3) (11.6 g, red solid) was obtained after drying in 96% yield. | | References | [1] Patent: US2006/173210, 2006, A1. Location in patent: Page/Page column 5 |
| | (S,S)-(+)-N,N'-BIS(3,5-DI-TERT-BUTYLSALICYLIDENE)-1,2-CYCLOHEXANEDIAMINO-COBALT(II) Preparation Products And Raw materials |
| Raw materials | Phenol, 2,2'-[(1S,2S)-1,2-cyclohexanediylbis(nitrilomethylidyne)]bis[4,6-bis(1,1-dimethylethyl)--->Cobalt acetate-->(S,S)-(+)-N,N'-BIS(3,5-DI-TERT-BUTYLSALICYLIDENE)-1,2-CYCLOHEXANEDIAMINE |
|