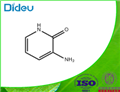
- 3-Amino-2(1H)-pyridinone
-
- $1.10 / 1g
-
2025-11-18
- CAS:33630-99-8
- Min. Order: 1g
- Purity: 99.00%
- Supply Ability: 100 Tons Min
- 3-Amino-2-pyridinol
-

- $1.00 / 1KG
-
2019-07-14
- CAS:33630-99-8
- Min. Order: 1KG
- Purity: 90%-99.9%
- Supply Ability: 500kg
|
| | 3-Amino-2(1H)-pyridinone Basic information | | Uses |
| | 3-Amino-2(1H)-pyridinone Chemical Properties |
| Melting point | 118-130 °C | | Boiling point | 206.4°C (rough estimate) | | density | 1.2111 (rough estimate) | | refractive index | 1.4800 (estimate) | | storage temp. | under inert gas (nitrogen or Argon) at 2–8 °C | | form | Powder | | pka | 13.87±0.10(Predicted) | | color | Dark brown to black | | Water Solubility | Slightly soluble in water. | | InChI | InChI=1S/C5H6N2O/c6-4-2-1-3-7-5(4)8/h1-3H,6H2,(H,7,8) | | InChIKey | VTSFNCCQCOEPKF-UHFFFAOYSA-N | | SMILES | C1(=O)NC=CC=C1N | | CAS DataBase Reference | 33630-99-8(CAS DataBase Reference) |
| Hazard Codes | Xi,Xn | | Risk Statements | 36/37/38-20/21/22 | | Safety Statements | 37/39-26-36 | | WGK Germany | WGK 3 | | HazardClass | IRRITANT | | HS Code | 29333999 | | Storage Class | 11 - Combustible Solids | | Hazard Classifications | Acute Tox. 4 Oral
Eye Dam. 1
Skin Irrit. 2
STOT SE 3 |
| Provider | Language |
|
ACROS
| English |
| | 3-Amino-2(1H)-pyridinone Usage And Synthesis |
| Uses | 3-Amino-2-hydroxypyridine has been used as a reactant in the preparation of benzothiazolyl hydroxyindolinylphenyl ureas as potent P2Y1 antagonists. | | Chemical Properties | Brown to off-white crystalline powder. | | Uses | It is used as pharmaceutical intermediate. It is involved in synthesis of functionalized pyrido[4,3-b][1,4]oxazine and imidazo[1,2-a]pyridine derivatives. The use of ethyl 2-chloro-3-oxopropanoate with 2-amino-3-hydroxypyridine or 3-amino-4-hydroxypyridine led, respectively, to imidazo[1,2-a]pyridine derivatives or ethyl pyrido[4,3-b][1,4] oxazine-2-carboxylate. | | Synthesis | a) Synthesis of 3-aminopyridin-2-ol: In a 500 mL three-necked flask, 3-nitropyridin-2(1H)-one (1 g, 7.14 mmol) was dissolved in methanol (200 mL), followed by addition of 10% palladium/activated carbon catalyst (100 mg, 10 wt%). The reaction mixture was stirred for 3 h under hydrogen atmosphere (1 atm) and at room temperature (25°C). Upon completion of the reaction, the catalyst was removed by filtration through a diatomaceous earth pad, and the filtrate was concentrated under reduced pressure by rotary evaporator to afford the white solid product 3-aminopyridin-2-ol (800 mg, quantitative yield). Product characterization data: 1H-NMR (CD3OD, 300 MHz) δ 6.78-6.73 (m, 2H), 6.23 (t, J = 6.5 Hz, 1H); MS (ESI) m/z 111.1 [M+H]+. | | References | [1] Patent: US2011/28467, 2011, A1. Location in patent: Page/Page column 14
[2] Patent: WO2006/51410, 2006, A1. Location in patent: Page/Page column 46
[3] Heterocycles, 1990, vol. 31, # 12, p. 2201 - 2204
[4] Recueil des Travaux Chimiques des Pays-Bas, 1949, vol. 68, p. 1013,1024 |
| | 3-Amino-2(1H)-pyridinone Preparation Products And Raw materials |
|