- Keto-pantoyllactone
-

- $29.00 / 200mg
-
2026-05-11
- CAS:13031-04-4
- Min. Order:
- Purity: 98.62%
- Supply Ability: 10g
|
| | DIHYDRO-4,4-DIMETHYL-2,3-FURANDIONE Basic information | | Uses |
| Product Name: | DIHYDRO-4,4-DIMETHYL-2,3-FURANDIONE | | Synonyms: | dihydro-4,4-dimethylfuran-2,3-dione;DIHYDRO-4,4-DIMETHYL-2,3-FURANDIONE;keto-pantoyllactone;4,4-Dimethyl-4,5-dihydro-2,3-furandione;4,4-Dimethyl-4,5-dihydrofuran-2,3-dione;4,4-Dimethyloxolane-2,3-dione;4,4-Dimethyltetrahydrofuran-2,3-dione;Ketopantolactone | | CAS: | 13031-04-4 | | MF: | C6H8O3 | | MW: | 128.13 | | EINECS: | 235-891-7 | | Product Categories: | API intermediates;Building Blocks;Furans;Heterocyclic Building Blocks | | Mol File: | 13031-04-4.mol | 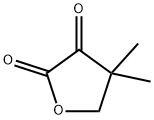 |
| | DIHYDRO-4,4-DIMETHYL-2,3-FURANDIONE Chemical Properties |
| Melting point | 67-69 °C (lit.) | | Boiling point | 125-126℃ (15 Torr) | | density | 1.152±0.06 g/cm3(Predicted) | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | solubility | dichloromethane: soluble25mg/mL, clear, colorless to yellow | | form | Solid | | color | Pale Beige | | InChI | InChI=1S/C6H8O3/c1-6(2)3-9-5(8)4(6)7/h3H2,1-2H3 | | InChIKey | HRTOQFBQOFIFEE-UHFFFAOYSA-N | | SMILES | O1CC(C)(C)C(=O)C1=O |
| WGK Germany | 3 | | Storage Class | 11 - Combustible Solids |
| | DIHYDRO-4,4-DIMETHYL-2,3-FURANDIONE Usage And Synthesis |
| Uses | Dihydro-4,4-dimethyl-2,3-furandione is a heterocyclic organic compound, mainly used as a pharmaceutical intermediate. | | Uses | Dihydro-4,4-dimethyl-2,3-furandione is an activated keto compound and its enantioselective hydrogenation was reported. Neutral Rhodium (I) aminophosphine-phosphinite complex calatyzed asymmetric hydrogenation of dihydro-4,4-dimethyl-2,3-furandione was reported. Asymmetric hydrogenation of dihydro-4,4-dimethyl-2,3-furandione gives D-(-)-pantoyl lactone, a key intermediate in the synthesis of pantothenic acid. | | Definition | ChEBI: 2-dehydropantolactone is a tetrahydrofurandione. It is functionally related to a pantoic acid. | | Synthesis Reference(s) | Synthetic Communications, 14, p. 697, 1984 DOI: 10.1080/00397918408063756 | | General Description | Dihydro-4,4-dimethyl-2,3-furandione is an activated keto compound and its enantioselective hydrogenation was reported. Neutral Rhodium (I) aminophosphine-phosphinite complex calatyzed asymmetric hydrogenation of dihydro-4,4-dimethyl-2,3-furandione was reported. Asymmetric hydrogenation of dihydro-4,4-dimethyl-2,3-furandione gives D-(-)-pantoyl lactone, a key intermediate in the synthesis of pantothenic acid. | | Synthesis | The general procedure for the synthesis of dihydro-4,4-dimethyl-2,3-furandione from DL-pantolactone was carried out as follows: 29.5 g of DL-pantolactone, 300 mg of ruthenium(III) chloride were added to a mixture of 29.5 g of DL-pantolactone, 300 mg of ruthenium(III) chloride in a microwave reactor (Ethos 1600, MLS GmbH, D-88299 Leutkirch im Allgau, Germany), A mixture of 400 mL of water and 200 mL of ethyl acetate was heated to reflux temperature with stirring and the microwave power was set to 700 W. Over a period of 10 min, 145.5 g of sodium meta-periodate was added to the boiling mixture in batches. The reaction mixture continued to be stirred for 20 min and then cooled rapidly. The organic and aqueous phases were separated using a separatory funnel and the sodium iodate precipitate in the organic phase was removed by diafiltration and washed five times with 20 mL of ethyl acetate. The aqueous phase was extracted twice with 50 mL of ethyl acetate. All organic phases were combined, dried with anhydrous magnesium sulfate, filtered and concentrated under reduced pressure. This step yields ketolactone with 98% purity and 60% yield based on starting DL-pantolactone. An additional 12% of the product as well as unreacted DL-pantolactone is recovered from the aqueous phase. The conversion of DL-pantolactone under these process conditions was 80% with a selectivity of 0.95. | | References | [1] Synthetic Communications, 1984, vol. 14, # 7, p. 697 - 700
[2] Patent: JP2005/75784, 2005, A. Location in patent: Page/Page column 20-21; 24-25
[3] Organic Process Research and Development, 2004, vol. 8, # 6, p. 931 - 938
[4] Russian Journal of Applied Chemistry, 2003, vol. 76, # 8, p. 1315 - 1318
[5] Synthetic Communications, 2002, vol. 32, # 1, p. 31 - 36 |
| | DIHYDRO-4,4-DIMETHYL-2,3-FURANDIONE Preparation Products And Raw materials |
|