- Cbz-L-Ser-Ome
-
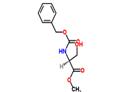
- $1.00 / 1g
-
2020-01-06
- CAS:1676-81-9
- Min. Order: 1g
- Purity: 98%
- Supply Ability: 100KG
|
| | N-Cbz-L-serine methyl ester Basic information |
| | N-Cbz-L-serine methyl ester Chemical Properties |
| Melting point | 41-43 °C(lit.) | | Boiling point | 170 °C0.01 mm Hg(lit.) | | density | 1.2499 (rough estimate) | | refractive index | -15 ° (C=1, MeOH) | | Fp | >230 °F | | storage temp. | Keep in dark place,Sealed in dry,Room Temperature | | solubility | Chloroform, Methanol | | pka | 10.49±0.46(Predicted) | | form | Viscous Liquid or Solid | | color | Pale yellow | | Optical Rotation | -13.4°(C=0.01 g/ml MEOH) | | CAS DataBase Reference | 1676-81-9(CAS DataBase Reference) |
| Safety Statements | 24/25 | | WGK Germany | 3 | | HS Code | 29242990 |
| | N-Cbz-L-serine methyl ester Usage And Synthesis |
| Chemical Properties | Clear Waxy Solid | | Uses | N-Cbz-L-serine methyl ester is used to inhibit the activity of Mycobacterium tuberculosis in studies. Also used in the synthesis of pyrazinecarboxamide-based compounds acting as inhibitors of diacylglycerol acetyltransferases regarding treatment of obesity. | | Uses | N-[(Phenylmethoxy)carbonyl]-L-serine Methyl Ester is used to inhibit the activity of Mycobacterium tuberculosis in studies. Also used in the synthesis of pyrazinecarboxamide-based compounds acting as inhibitors of diacylglycerol acetyltransferases regarding treatment of obesity. | | reaction suitability | reaction type: solution phase peptide synthesis | | Synthesis | To a suspension of L-serine methyl ester hydrochloride (1.00 g, 6.43 mmol, 1.00 equiv) in dioxane (5.00 mL) was added an aqueous solution (25.7 mL) of NaHCO3 (1.62 g, 19.3 mmol, 3.00 equiv). Subsequently, a dioxane solution (15.0 mL, diluted with 4.20 mL dioxane) of benzyl chloroformate (Cbz-Cl, 1.10 mL, 7.72 mmol, 1.20 eq.) was added to the reaction system and the reaction was stirred at 25 °C. After 5 hours of reaction, the reaction was quenched with 1 M HCl aqueous solution and the aqueous layer was extracted with ethyl acetate (2×). The organic layers were combined, washed with 10% NaCl aqueous solution, dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The crude product was purified by silica gel column chromatography (eluent: hexane solution of 50% ethyl acetate) to afford N-benzyloxycarbonyloxy-L-serine methyl ester (1.50 g, 5.92 mmol, 92% yield) as a colorless oil. | | References | [1] Journal of Organic Chemistry, 1996, vol. 61, # 16, p. 5528 - 5531
[2] Organic and Biomolecular Chemistry, 2015, vol. 13, # 26, p. 7177 - 7192
[3] Synthesis, 1996, # 2, p. 189 - 191
[4] Beilstein Journal of Organic Chemistry, 2017, vol. 13, p. 106 - 110
[5] Organic Letters, 2018, vol. 20, # 4, p. 1019 - 1022 |
| | N-Cbz-L-serine methyl ester Preparation Products And Raw materials |
|