Copper(II) sulfate pentahydrate
- CAS No.
- 7758-99-8
- Chemical Name:
- Copper(II) sulfate pentahydrate
- Synonyms
- COPPER SULPHATE PENTAHYDRATE;malachite;Copper (II) sulfate;Copper(Ⅱ) sulfate pentahydrate;Copper (II) sulfate pentahydrate (as Cu);csp;CUPRIC SULFATE, ACS;Copper sulfate,medicinal;COPPER(II) SULPHATE PENTAHYDRATE;Copper(II)Sulfatepentahydrate,99.995%
- CBNumber:
- CB6705282
- Molecular Formula:
- CuH10O9S
- Molecular Weight:
- 249.68
- MDL Number:
- MFCD00149681
- MOL File:
- 7758-99-8.mol
- MSDS File:
- SDS
| Product description | Number | Pack Size | Price |
| Copper(II) sulfate solution 4 % (w/v) (prepared from copper (II) sulfate pentahydrate) | C2284 | 25 mL | $47.6 |
| Copper(II) sulfate pentahydrate purum p.a., crystallized, ≥99.0% (RT) | 61245 | 500g | $124 |
| Copper(II) sulfate pentahydrate ≥99.9% (trace metals basis) | 939315 | 1 unit | $105 |
| Copper(II) sulfate pentahydrate purum p.a., crystallized, ≥99.0% (RT) | 61245 | 1kg | $182 |
| Copper(II) sulfate pentahydrate purum p.a., crystallized, ≥99.0% (RT) | 61245 | 6x1kg | $1040 |
| More product size | |||
| Melting point | 110 °C (dec.)(lit.) |
|---|---|
| Density | 2.284 |
| vapor pressure | 7.3 mm Hg ( 25 °C) |
| refractive index | 1.537 |
| storage temp. | Store at +5°C to +30°C. |
| solubility | 320 g/L (20°C) |
| form | Solid |
| color | fine blue crystals |
| Specific Gravity | 2.284 |
| PH | 3.5-4.5 (25℃, 50mg/mL in H2O) |
| Odor | blue cryst. or cryst. gran. or powd., odorless |
| Water Solubility | 320 g/L (20 ºC) |
| Merck | 14,2653 |
| Exposure limits |
ACGIH: TWA 1 mg/m3 NIOSH: IDLH 100 mg/m3; TWA 1 mg/m3 |
| InChI | 1S/Cu.H2O4S.5H2O/c;1-5(2,3)4;;;;;/h;(H2,1,2,3,4);5*1H2/q+2;;;;;;/p-2 |
| InChIKey | JZCCFEFSEZPSOG-UHFFFAOYSA-L |
| SMILES | O.O.O.O.O.[Cu++].[O-]S([O-])(=O)=O |
| CAS DataBase Reference | 7758-99-8(CAS DataBase Reference) |
| EWG's Food Scores | 1 |
| NCI Dictionary of Cancer Terms | CSP |
| FDA UNII | LRX7AJ16DT |
| EPA Substance Registry System | Copper sulfate pentahydrate (7758-99-8) |
| Pesticides Freedom of Information Act (FOIA) | Copper sulfate pentahydrate |
| UNSPSC Code | 51211624 |
| NACRES | NA.71 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS05,GHS07,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H302-H318-H410 | |||||||||
| Precautionary statements | P264-P270-P273-P280-P301+P312-P305+P351+P338 | |||||||||
| Hazard Codes | Xn,N,Xi | |||||||||
| Risk Statements | 22-36/38-50/53-52/53-36-36/37/38-20/21/22-51/53 | |||||||||
| Safety Statements | 22-60-61-26-36 | |||||||||
| RIDADR | UN 3288 6.1/PG 3 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | GL8900000 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 6.1 | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 28332500 | |||||||||
| Storage Class | 13 - Non Combustible Solids | |||||||||
| Hazard Classifications | Acute Tox. 4 Oral Aquatic Acute 1 Aquatic Chronic 1 Eye Dam. 1 |
|||||||||
| Hazardous Substances Data | 7758-99-8(Hazardous Substances Data) | |||||||||
| Toxicity | LD50 orally in rats: 960 mg/kg (Smyth) | |||||||||
| Limited Quantities | 5.0 L (1.3 gallons) (liquid) or 5.0 kg (11 lbs) (solid) | |||||||||
| Excepted Quantities | Max Inner Pack (30g or 30ml) and Max Outer Pack (1Kg or 1L) | |||||||||
| NFPA 704 |
|
Copper(II) sulfate pentahydrate price More Price(122)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | C2284 | Copper(II) sulfate solution 4 % (w/v) (prepared from copper (II) sulfate pentahydrate) | 7758-99-8 | 25 mL | $47.6 | 2025-07-31 | Buy |
| Sigma-Aldrich | 61245 | Copper(II) sulfate pentahydrate purum p.a., crystallized, ≥99.0% (RT) | 7758-99-8 | 500g | $124 | 2025-07-31 | Buy |
| Sigma-Aldrich | 939315 | Copper(II) sulfate pentahydrate ≥99.9% (trace metals basis) | 1 unit | $105 | 2025-07-31 | Buy | |
| Sigma-Aldrich | 61245 | Copper(II) sulfate pentahydrate purum p.a., crystallized, ≥99.0% (RT) | 7758-99-8 | 1kg | $182 | 2025-07-31 | Buy |
| Sigma-Aldrich | 61245 | Copper(II) sulfate pentahydrate purum p.a., crystallized, ≥99.0% (RT) | 7758-99-8 | 6x1kg | $1040 | 2025-07-31 | Buy |
Copper(II) sulfate pentahydrate Chemical Properties,Uses,Production
Description
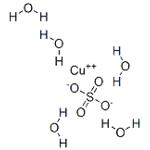
Copper(II) sulfate pentahydrate is known as blue vitriol. It is an odorless blue crystal that readily dissolves in water. It is also soluble in methanol, glycerol and slightly soluble in ethanol. The highly toxic, non-combustible has a nauseating metallic taste and turns white when dehydrated. Structurally, in the pentahydrate molecule, each copper(II) ions is surrounded by four water molecules in the corners and the fifth water molecule is attached by hydrogen bonding. Copper (II) sulfate has many applications including preparation of Bordeaux mixture, a fungicide preparation. Electroplating, timber preservation and textile industry use copper (II) sulfate.
Chemical Properties
Copper sulfate is a greenish-white crystalline solid; the pentahydrate is Blue powder or granules, or ultramarine crystalline solid.
Uses
Used as a source of Cu2+ ions
Uses
Anhydr salt for detecting and removing trace amounts of water from alcohols and other organic Compounds; as fungicide. Pentahydrate as agricultural fungicide, algicide, bactericide, herbicide; food and fertilizer additive; in insecticide mixtures; in manufacture of other Cu salts; as mordant in textile dyeing; in preparation of azo dyes; in preserving hides; in tanning leather; in preserving wood; in electroplating solutions; as battery electrolyte; in laundry and metal-marking inks; in petroleum refining; as flotation agent; pigment in paints, varnishes and other materials; in mordant baths for intensifying photographic negatives; in pyrotechnic compositions; in water-resistant adhesives for wood; in metal coloring and tinting baths; in antirusting compositions for radiator and heating systems; as reagent toner in photography and photoengraving; etc.
Uses
Copper (II) sulfate pentahydrate salt may be used for the fabrication of copper nanoparticles by chemical reduction. The pentahydrate salt of copper may be used as a catalyst for the conversion of aromatic aldehydes to primary amides via aldoximes. Reduced graphene oxide-supported copper nanoparticles (rGO/Cu NPs) may be prepared by copper (II) sulfate pentahydrate and graphitic precursors. An aqueous electrolytic bath containing CuSO4.5H2O as one of the constituents was used for the preparation of Cu2ZnSnS4 (CZTS) thin film solar cell. Ferric chloride hexahydrate (FeCl3.6H2O) and copper(II) sulfate pentahydrate (CuSO4.5H2O) may be used to fabricate Fe-Cu binary oxide sorbents for arsenic removal applications.
Application
Copper(II) sulfate pentahydrate has been used:
as an additive in trace element solution preparation in solid glucose minimal media.
as a component of adamsII solution in Pneumococcal media.
in the preparation of alginate gel for drug encapsulation.
Copper(II) sulfate is an inorganic Lewis acid commonly used to promote acid catalyzed organic reactions. It is used as a reagent for the synthesis of copper carbenoids. It can also act as an effective redox catalyst in combination with other mixed oxidizing systems.
Copper Sulfate Pentahydrate Fine Crystals serve as the main ingredient for manufacturing Bordeaux and Burgundy mixtures that are used as algaecides, both on the farm and to ensure safe water supplies. Bordeaux mixtures are also employed to adjust and maintain copper deficient soils to optimum levels.
Definition
A green mineral consisting of copper(II) carbonate and hydroxide (CuCO3.Cu(OH)2). It is used as an ore and a pigment.
Definition
malachite: Apresumptive test for blood. Thereagent is the dye leucomalachitegreen dissolved in water along withsodium perborate (NaBO3). A bluegreencolour indicates a positive result.
Definition
ChEBI: Copper(II) sulfate pentahydrate is the pentahydrate of copper(2+) sulfate. A bright blue crystalline solid. It is a hydrate and a metal sulfate. It contains a copper(II) sulfate.
General Description
Blue crystalline granules or powder. Melting point 110°C (with decomposition). Non-combustible. Nauseating metallic taste. Odorless. White when dehydrated.
Air & Water Reactions
Slowly effloresces in air. Water soluble.
Reactivity Profile
Copper sulfate pentahydrate can be dehydrated by heating. Serves as a weak oxidizing agent. Causes hydroxylamine to ignite. Gains water readily. The hydrated salt is vigorously reduced by hydroxylamine [Mellor 8:292(1946-1947)]. Both forms are incompatible with finely powdered metals. Both are incompatible with magnesium, corrode steel and iron, may react with alkalis, phosphates, acetylene gas, hydrazine, or nitromethane, and may react with beta-naphthol, propylene glycol, sulphathiazole and triethanolamine if the pH exceeds 7 . Both act as acidic salts, corrode metals and irritate tissues.
Fire Hazard
Literature sources indicate that Copper sulfate pentahydrate is nonflammable.
reaction suitability
reagent type: catalyst
core: copper
Potential Exposure
Copper sulfate is used as intermediate and wood preservative; also used in production of copper compounds; to detect and to remove trace amounts of water from alcohols and organic compounds; as a fungicide and algicide; in veterinary medicine and others.
Shipping
UN3288 Toxic solids, inorganic, n.o.s., Hazard Class: 6.1; Labels: 6.1-Poisonous materials, Technical Name Required. UN3077 Environmentally hazardous substances, solid, n.o.s., Hazard class: 9; Labels: 9-Miscellaneous hazardous material, Technical Name Required.
Incompatibilities
Aqueous solution is an acid. May form explosive materials on contact with acetylene and nitromethane. Incompatible with strong bases; hydroxylamine, magnesium; zirconium, sodium hypobromite, hydrazine.
Toxics Screening Level
The screening levels for both of the above-mentioned copper compounds are 10 μg/m 3 based on 8 hour averaging.
Waste Disposal
Copper-containing soluble wastes can be concentrated through the use of ion exchange, reverse osmosis, or evaporators to the point where copper can be electrolytically removed and sent to a reclaiming firm. If recovery is not feasible, the copper can be precipitated through the use of caustics and the sludge deposited in a chemical waste landfill Add soda ash to waste CuSO4 solution; let stand 24 hours. Decant and neutralize solution before flushing to sewer. Landfill sludge.
Copper(II) sulfate pentahydrate Preparation Products And Raw materials
Raw materials
1of3
Preparation Products
1of6
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Hebei Andu Technology Com.,Ltd | +86-86-17798073498 +8617798073498 | aduchemical@163.com | China | 296 | 58 |
| Hebei Chuanghai Biotechnology Co., Ltd | +8615350571055 | Sibel@chuanghaibio.com | China | 8749 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +8617392709771 | 1097@dideu.com | China | 3995 | 58 |
| Qingdao Trust Agri Chemical Co.,Ltd | +86-008615963231493 +86-008613573296305 | trustagri@hotmail.com | China | 301 | 58 |
| Wuhan Fortuna Chemical Co., Ltd | +86-027-59207850 +86-13986145403; | info@fortunachem.com | China | 6010 | 58 |
| Hebei Chuanghai Biotechnology Co,.LTD | +86-86-13131129325 +8613131129325 | sales1@chuanghaibio.com | China | 5240 | 58 |
| Shaanxi TNJONE Pharmaceutical Co., Ltd | +8618092446649 | sarah@tnjone.com | China | 1143 | 58 |
| Lianyungang Kaiyu Environmental Tech Co., Ltd. | +86-0086-19851820538 +8619851820538 | carlos@khonorchem.com | China | 314 | 58 |
| Jiangsu Kolod Food Ingredients Co.,Ltd. | +8618805133257 | sales3257@jskolod.com | China | 134 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21595 | 55 |
Related articles
- Malachite synthesis
- Chemically, Malachite is a combination of copper(II) carbonate and copper(II) hydroxide, CuCO3xCu(OH)2 .
- Jul 31,2024
View Lastest Price from Copper(II) sulfate pentahydrate manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2026-03-13 | Copper sulfate pentahydrate
7758-99-8
|
0.99 | RongNa Biotechnology Co.,Ltd | ||||
 |
2026-03-13 | Copper(II) sulfate pentahydrate
7758-99-8
|
US $0.00-0.00 / kg | 1kg | 99.5% | 1000kg | WUHAN FORTUNA CHEMICAL CO., LTD | |
 |
2026-03-06 | Copper sulfate pentahydrate
7758-99-8
|
US $10.00 / KG | 100KG | 99% | 100 mt | Hebei Chuanghai Biotechnology Co., Ltd |
-

- Copper sulfate pentahydrate
7758-99-8
- 0.99
- RongNa Biotechnology Co.,Ltd
-

- Copper(II) sulfate pentahydrate
7758-99-8
- US $0.00-0.00 / kg
- 99.5%
- WUHAN FORTUNA CHEMICAL CO., LTD
-

- Copper sulfate pentahydrate
7758-99-8
- US $10.00 / KG
- 99%
- Hebei Chuanghai Biotechnology Co., Ltd





