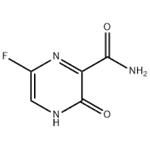
Favipiravir
- CAS No.
- 259793-96-9
- Chemical Name:
- Favipiravir
- Synonyms
- 6-Fluoro-3-hydroxypyrazine-2-carboxamide;T-705;Favipiravi;6-fluoro-3,4-dihydro-3-oxo-Pyrazinecarboxamide;Avigan;Farapiwe;3-Dithian;Fapilawei;T-705,T705;3-Dithiane
- CBNumber:
- CB71055246
- Molecular Formula:
- C5H4FN3O2
- Molecular Weight:
- 157.1
- MDL Number:
- MFCD12032148
- MOL File:
- 259793-96-9.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| T-705 ≥98% | 23384 | 10mg | $38 |
| T-705 ≥98% | 23384 | 25mg | $52 |
| T-705 ≥98% | 23384 | 50mg | $94 |
| T-705 ≥98% | 23384 | 100mg | $129 |
| T-705 ≥98% | 23384 | 50mg | $85 |
| More product size | |||
| Melting point | >151°C (dec.) |
|---|---|
| Density | 1.78±0.1 g/cm3(Predicted) |
| storage temp. | under inert gas (nitrogen or Argon) at 2-8°C |
| solubility | Soluble in DMSO (30 mg/mL), water (12 mg/mL with warming) |
| pka | 8.77±0.60(Predicted) |
| form | solid |
| color | Off-white |
| Stability | Stable for 1 year from date of purchase as supplied. Solutions in DMSO or water may be stored at -20° for up to 3 months. |
| InChI | InChI=1S/C5H4FN3O2/c6-2-1-8-5(11)3(9-2)4(7)10/h1H,(H2,7,10)(H,8,11) |
| InChIKey | ZCGNOVWYSGBHAU-UHFFFAOYSA-N |
| SMILES | C1(C(N)=O)=NC(F)=CNC1=O |
| FDA UNII | EW5GL2X7E0 |
| ATC code | J05AX27 |
| UNSPSC Code | 41116107 |
| NACRES | NA.24 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H341-H361d | |||||||||
| Precautionary statements | P201-P202-P280-P308+P313-P405-P501 | |||||||||
| WGK Germany | WGK 3 | |||||||||
| Storage Class | 11 - Combustible Solids | |||||||||
| Hazard Classifications | Muta. 2 Repr. 2 |
|||||||||
| NFPA 704 |
|
Favipiravir price More Price(39)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Cayman Chemical | 23384 | T-705 ≥98% | 259793-96-9 | 10mg | $38 | 2026-04-30 | Buy |
| Cayman Chemical | 23384 | T-705 ≥98% | 259793-96-9 | 25mg | $52 | 2026-04-30 | Buy |
| Cayman Chemical | 23384 | T-705 ≥98% | 259793-96-9 | 50mg | $94 | 2026-04-30 | Buy |
| Cayman Chemical | 23384 | T-705 ≥98% | 259793-96-9 | 100mg | $129 | 2026-04-30 | Buy |
| Cayman Chemical | 23384 | T-705 ≥98% | 259793-96-9 | 50mg | $85 | 2024-03-01 | Buy |
Favipiravir Chemical Properties,Uses,Production
Description
Favipiravir, 6-fluoro-3-hydroxypyrazine-2-carboxamide, is a new broad-spectrum antiviral drug targeting RNA-dependent RNA polymerase (RdRp) developed by Japan's Toyama Chemical Pharmaceutical Company. It was approved for marketing in Japan in March 2014 for the treatment of new and recurrent influenza. During the outbreak of the new coronavirus, the results of the Phase I clinical study of the drug published in March 2020 showed that the drug may have the effect of speeding up virus clearance to alleviate the progress of new coronavirus pneumonia.
History
Favipiravir was originally developed in the late 1990s by a company that was later purchased by the Japanese firm Fujifilm as part of its transition from the photo business to healthcare. After being tested against a range of viruses, the drug was approved in Japan in 2014 for emergency use against flu epidemics or to treat new strains of influenza.
Uses
Favipiravir is used for the treatment of advanced Ebola virus infection in a small animal model. It suppressed the replication of Zaire Ebola virus and prevented a lethal outcome in 100% of the animals. Based on the studies, Favipiravir can be a candidate for the treatment of Ebola hemorrhagic fever. Favipiravir can be used for antiviral treatment of influenza A and B. Studies have shown that in addition to influenza virus, the drug also exhibits good antiviral activity against a variety of RNA viruses, such as Ebola virus, arena virus, Bunia virus, rabies virus and now COVID-19.
Definition
ChEBI: Favipiravir is a member of the class of pyrazines that is pyrazine substituted by aminocarbonyl, hydroxy and fluoro groups at positions 2, 3 and 6, respectively. It is an anti-viral agent that inhibits RNA-dependent RNA polymerase of several RNA viruses and is approved for the treatment of influenza in Japan. It has a role as an antiviral drug, an anticoronaviral agent and an EC 2.7.7.48 (RNA-directed RNA polymerase) inhibitor. It is a primary carboxamide, a hydroxypyrazine and an organofluorine compound.
Mechanism of action
Favipiravir is an antiviral drug that selectively inhibited the RdRP of influenza virus. It showed specific activity against all three influenza A, B, and C (Furuta et al., 2013). It also inhibited the RV replication in HeLa cells, with an EC50 of 29 μg/mL (Furuta et al., 2002). Analysis showed that the primary mechanism of action of favipiravir against the influenza virus was specific inhibition of vRNA polymerase (Furuta et al., 2005). It is predicted that a similar mechanism might occur with other viruses, such as PV and RV, inhibited by favipiravir, which may account for its broad-spectrum inhibition. Mechanistic studies show that the favipiravir and its form favipiravir-RMP (favipiravir-ribofuranosyl-50-monophosphate) do not inhibit influenza RNA polymerase activity, but it is the phosphoribosylated form, favipiravir-ribofuranosyl-50-triphosphate (RTP) that inhibits the enzyme.
Synthesis
Favipiravir was synthesized in only six steps from 3-aminopyrazine-2-carboxylic acid with an overall yield of about 22.3%. Key intermediates 3 and 6 were obtained in excellent purity via recrystallization from optimized solvents, which was beneficial to large-scale production. In the key synthetic reaction, 3,6-dichloropyrazine-2-carbonitrile (6) was reacted sequentially, in one pot, with KF and 30% H2O2 to give (after crystallization from 95% EtOH) favipiravir as colorless crystals, with a 60% yield for this final step of the synthesis. (DOI:10.1007/s11696-017-0208-6)
645 grams of sodium hydroxide were dissolved in 9L of water, the temperature was lowered to 5C, and 1.29 kg of 6-fluoro-3-hydroxy-2-cyanopyrazine was added in batches, stirred, and heated slightly, and the temperature of the reaction system was controlled to -10C, it takes 3.5 hours to complete the addition, after holding for 1 hour, the temperature is raised to 40C for 1 hour. Add 100g of activated carbon to the reaction solution, hot filter, cool the mother liquor to 5C, adjust the pH to 3-4 with concentrated hydrochloric acid, precipitate a large amount of solids, filter and dry to obtain a crude off-white powder, beaten with 2.8 liters of 15% methanol aqueous solution and filter After drying, 1.34 kg of white powder favipiravir was obtained. 1H-NMR (DMSO, 600MHz): | 13.38 (s, 1H), 8.73 (1s, 1H), 8.51-8.49 (d, J=12, 2H) (yield 91%).
storage
Store at -20°C
Background
T-705 is a potent antiviral agent that has shown effectiveness against influenzas A, B, and C with IC50 values as follows: A = 0.03-0.20 µg/ml, A = 0.01-0.30 µg/ml, A = 0.08-0.48 µg/ml, A = 0.14-0.15 µg/ml, A = 0.24-1.60 µg/ml, B = 0.04-0.09 µg/ml, and C = 0.03-0.06 µg/ml. T-705 becomes activated by phosphoribosylation to form T-705-RTP that is recognized by RNA polymerase, inhibiting RNA-dependent RNA polymerase. This activity makes T-705 an effective inhibitor of other RNA viruses, such as arena-, phlebo-, hanta-, flavi-, entero-, and alphavirus. The antiviral capability of T-705 makes it a compound of interest when studying SARS-CoV-2 infection.
References
[1] Y. FURUTA. In Vitro and In Vivo Activities of Anti-Influenza Virus Compound T-705[J]. Antimicrobial Agents and Chemotherapy, 2002, 46 1: 977-981. DOI:10.1128/aac.46.4.977-981.2002
[2] KAZUMI TAKAHASHI. In vitro and in vivo activities of T-705 and oseltamivir against influenza virus.[J]. Antiviral Chemistry and Chemotherapy, 2003, 14 5: 235-241. DOI:10.1177/095632020301400502
[3] KATRINA SLEEMAN. In vitro antiviral activity of favipiravir (T-705) against drug-resistant influenza and 2009 A(H1N1) viruses.[J]. Antimicrobial Agents and Chemotherapy, 2010, 54 6: 2517-2524. DOI:10.1128/aac.01739-09
[4] YOUSUKE FURUTA. Mechanism of action of T-705 against influenza virus.[J]. Antimicrobial Agents and Chemotherapy, 2005: 981-986. DOI:10.1128/aac.49.3.981-986.2005
[5] YOUSUKE FURUTA . Favipiravir (T-705), a novel viral RNA polymerase inhibitor[J]. Antiviral research, 2013, 100 2: Pages 446-454. DOI:10.1016/j.antiviral.2013.09.015
[6] LIYING DONG Jianjun G Shasha Hu. Discovering drugs to treat coronavirus disease 2019 (COVID-19).[J]. Drug Discoveries and Therapeutics, 2020, 14 1: 58-60. DOI:10.5582/ddt.2020.01012
[7] YUNG-FANG TU. A Review of SARS-CoV-2 and the Ongoing Clinical Trials.[J]. International Journal of Molecular Sciences, 2020, 21 7. DOI:10.3390/ijms21072657
Favipiravir Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | sales@conier.com | China | 49977 | 58 |
| Qiuxian Baitai New Material Co., LTD | +8618330912755 | sale2@hbyalin.com | China | 1658 | 58 |
| HEBEI Meijinnong Import And Export Trade Co.,LTD | +8617363028569 | admin@mjn-cn.com | China | 78 | 58 |
| Hebei Ningnan Trade Co. LTD | +86-18034019111 | admin@hbningnan.com | China | 255 | 58 |
| HangZhou RunYan Pharma Technology Co.,LTD. | +8618882027439 | sales2@runyanpharma.com | China | 302 | 58 |
| Hebei Dangtong Import and export Co LTD | +86-86-4001020630 +8619831957301 | admin@hbdangtong.com | China | 1000 | 58 |
| Hebei Chuanghai Biotechnology Co., Ltd | +8615350571055 | Sibel@chuanghaibio.com | China | 8738 | 58 |
| Hebei Yanxi Chemical Co., Ltd. | +8618531123677 | faithe@yan-xi.com | China | 5853 | 58 |
| XI'AN TIANGUANGYUAN BIOTECH CO., LTD. | +86-029-86333380 +86-18829239519 | sales06@tgybio.com | China | 898 | 58 |
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 | deasea125996@gmail.com | China | 2472 | 58 |
Related articles
- Favipiravir: A Repurposed Antiviral Drug Showing Promising COVID-19 Treatment
- Favipiravir shows promise in treating COVID-19 by inhibiting virus replication, with effectiveness dependent on a higher dosin....
- Feb 19,2024
- Favipiravir: mechanism of action, pharmacokinetics and safety
- Favipiravir is a prodrug that inhibits RNA polymerase, disrupts viral replication, and increases mutations. It has broad antiv....
- Sep 22,2023
- What is Favipiravir?
- Favipiravir is a pyrazine carboxamide derivative developed by Toyama Chemical of Japan to act against many RNA viruses. It was....
- Jul 21,2020
View Lastest Price from Favipiravir manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2026-05-02 | Pyrazinecarboxamide, 6-fluoro-3,4-dihydro-3-oxo- (9CI)
259793-96-9
|
0.99 | RongNa Biotechnology Co.,Ltd | ||||
 |
2026-04-30 | Favipiravir
259793-96-9
|
US $0.00 / Kg/Bag | 10g | 99%min HPLC | 10KGS | WUHAN FORTUNA CHEMICAL CO., LTD | |
 |
2026-04-30 | Favipiravir
259793-96-9
|
US $0.00 / g | 1g | More Than 99% | 100kg/Month | BEIJING SJAR TECHNOLOGY DEVELOPMENT CO., LTD. |
-

- Pyrazinecarboxamide, 6-fluoro-3,4-dihydro-3-oxo- (9CI)
259793-96-9
- 0.99
- RongNa Biotechnology Co.,Ltd
-

- Favipiravir
259793-96-9
- US $0.00 / Kg/Bag
- 99%min HPLC
- WUHAN FORTUNA CHEMICAL CO., LTD
-

- Favipiravir
259793-96-9
- US $0.00 / g
- More Than 99%
- BEIJING SJAR TECHNOLOGY DEVELOPMENT CO., LTD.







