|
|
| | 6-(Benzyloxy)-9-((1S,3S)-4-(benzyloxy)-3-((benzyloxy)methyl)-2-methylenecyclopentyl)-N-((4-methoxyphenyl)diphenylmethyl)-9H-purin-2-amine Basic information |
| Product Name: | 6-(Benzyloxy)-9-((1S,3S)-4-(benzyloxy)-3-((benzyloxy)methyl)-2-methylenecyclopentyl)-N-((4-methoxyphenyl)diphenylmethyl)-9H-purin-2-amine | | Synonyms: | 6-(Benzyloxy)-9-(1S,3R,3S)-4-(benzyloxy)-3-(benzyoxymethyl)-2-methylenecyclopentyl)-N-N-(4-methyoxyphenyl)diaphenylmethyl)-9H-purin-2-amine;6-(benzyloxy)-9-(1S,3R,3S)-4-(benzyloxy)_3-(benzyoxymethyl)-2-methylenecyclopentyl)-N-N
-(4-methyoxyphenyl)diaphenylmethyl)-9H-purin-2-amino;6-(Benzyloxy)-9-((1S,3R,3S)-4-(benzyloxy)-3-(benzyloxymethyl)-2-methylenecyclopentyl)-N-((4-methoxyphenyl)diphenylmethyl)-9H-purin-2-amine;[1S-(1α,3α,4β)]-N-[(4-Methoxyphenyl)diphenyl-Methyl]-6-(phenylMethoxy)-9-[2-Methylene-4-(phenylMethoxy)-3-[(phenylMethoxy)Methyl]-cyclopentyl]-9H-purin-2-aMine;Entecavir InterMediate VII;N-[(4-Methoxyphenyl)diphenylMethyl]-9-[(1S,3R,4S)-2-Methylene-4-(benzyloxy)-3-[(benzyloxy)Methyl]cyclopentyl]-6-(benzyloxy)-9H-Purin-2-aMine;Entecavir-7;Entecavir interMediate 7 | | CAS: | 142217-80-9 | | MF: | C53H49N5O4 | | MW: | 819.99 | | EINECS: | 1806241-263-5 | | Product Categories: | | | Mol File: | 142217-80-9.mol | 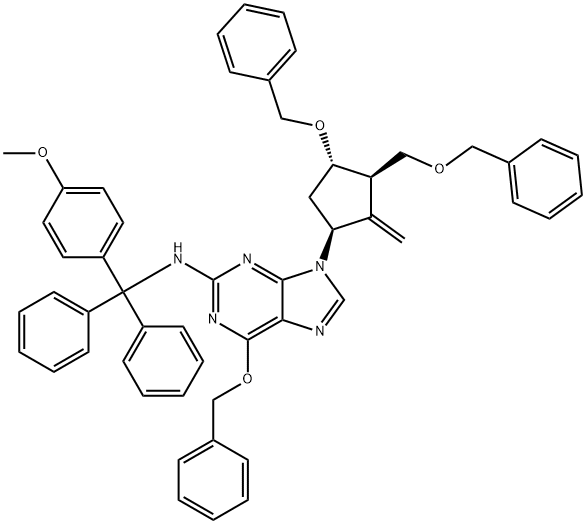 |
| | 6-(Benzyloxy)-9-((1S,3S)-4-(benzyloxy)-3-((benzyloxy)methyl)-2-methylenecyclopentyl)-N-((4-methoxyphenyl)diphenylmethyl)-9H-purin-2-amine Chemical Properties |
| density | 1.19 | | storage temp. | Keep in dark place,Inert atmosphere,Room temperature | | pka | 3.08±0.30(Predicted) | | InChIKey | OXHGMTPGAFKZHK-QQVWZEJNSA-N | | SMILES | N1C2C(=NC(NC(C3=CC=C(OC)C=C3)(C3=CC=CC=C3)C3=CC=CC=C3)=NC=2OCC2=CC=CC=C2)N([C@H]2C[C@H](OCC3=CC=CC=C3)[C@@H](COCC3=CC=CC=C3)C2=C)C=1 | | CAS DataBase Reference | 142217-80-9(CAS DataBase Reference) |
| | 6-(Benzyloxy)-9-((1S,3S)-4-(benzyloxy)-3-((benzyloxy)methyl)-2-methylenecyclopentyl)-N-((4-methoxyphenyl)diphenylmethyl)-9H-purin-2-amine Usage And Synthesis |
| Chemical Properties | White powder | | Uses | 6-(Benzyloxy)-9-((1S,3S)-4-(benzyloxy)-3-((benzyloxy)methyl)-2-methylenecyclopentyl)-N- ((4-methoxyphenyl)diphenylmethyl)-9H-purin-2-amine is an intermediate compound in the synthesis of the antiviral drug Entecavir. It can be used as a reference standard reagent for the quality control and analysis of Entecavir. | | Definition | 6-(Benzyloxy)-9-((1S,3S)-4-(benzyloxy)-3-((benzyloxy)methyl)-2-methylenecyclopentyl)-N-((4-methoxyphenyl)diphenylmethyl)-9H-purin-2-amine is also known as Entecavir Impurity 3. Entecavir belongs to the family of medicines called antivirals. Entecavir is used to treat liver infections caused by hepatitis B virus. | | Synthesis | Step (1) Preparation of Intermediate IX: In a 20L sealed and nitrogen-filled reaction flask, 3.8kg of zinc powder and 10wt% hydrochloric acid (12.5L) were added sequentially and stirred at room temperature for 10-15 minutes, followed by diafiltration, and washed with purified water to neutral. Next, it was washed with 3.0 L of ethanol and 4.5 L of tetrahydrofuran and dried under vacuum at 40±2°C for 4-4.5 hours to obtain about 3 kg of activated zinc powder. The activated zinc powder needs to be sealed and stored under vacuum for backup. In a 100L reactor, after evacuation and replacement of nitrogen, add 40L of tetrahydrofuran dried by A-type molecular sieve. Under stirring, 3 kg of dry activated zinc powder and 1.3 L of dibromomethane were added, and the mixture was cooled to -70~-55°C. Then 1140 ml of titanium tetrachloride was added dropwise with continuous stirring for 1 hour. The temperature was then increased and the reaction was carried out for 48±0.5 h. The reaction was kept sealed during the reaction. The prepared enolization reagent was cooled to -20°C and a solution of 1.2 kg of Intermediate VIII dissolved in 20 L of dichloromethane was slowly added dropwise at -30°C. After the dropwise addition, the temperature was raised to 15-20 °C and the reaction was continued with stirring for 180 ± 10 min. The end point of the reaction was monitored by TLC. The reaction solution was transferred to a 200 L reaction vessel (containing a mixed system of 30 L of dichloromethane, 100 L of pure water and 10 kg of sodium bicarbonate) and stirred for 60-70 min. The mixture was separated using a stainless steel centrifuge and the filter cake was washed twice with dichloromethane (16L). The filtrates were combined and the aqueous phase was subjected to secondary extraction with dichloromethane (16L x 2). The combined organic phases were concentrated to dryness under vacuum at 25-30°C to afford 6-benzyloxy-9-((1S,3R,3S)-4-benzyloxy-3-benzyloxymethyl-2-methylenecyclopentyl)-N-((4-methoxyphenyl)diphenylmethyl)-9H-purin-2-amine (Intermediate IX) as a yellow powder in a single-step yield of 88% and purity of 93.6%. | | References | [1] Patent: CN108148061, 2018, A. Location in patent: Paragraph 0028; 0029; 0030; 0031; 0032; 0033; 0034 |
| | 6-(Benzyloxy)-9-((1S,3S)-4-(benzyloxy)-3-((benzyloxy)methyl)-2-methylenecyclopentyl)-N-((4-methoxyphenyl)diphenylmethyl)-9H-purin-2-amine Preparation Products And Raw materials |
| Raw materials | 4-Methoxytriphenylmethyl chloride-->Benzene, [(2,4-cyclopentadien-1-ylmethoxy)methyl]--->(2R,3S,5S)-3-(Benzyloxy)-5-[2-[[(4-methoxyphenyl)diphenylmethyl]amino]-6-(benzyloxy)-9H-purin-9-yl]-2-(benzyloxymethyl)cyclopentanone-->(1S,2R,3S,5R)-2-(Benzyloxymethyl)-6-oxabicyclo[3.1.0]hexan-3-ol-->SODIUM CYCLOPENTADIENIDE-->(1S,2R,3S,5R)-3-(Phenymethyloxy)-2-(phenylmethoxy)methyl-6-oxabicyclo[3.1.0]hexane-->(1S, 2R)-2-(Benzyloxymethyl)-1-hydroxy-3-cyclopentene-->(2R,3S,5S)-3-(Benzyloxy)-5-[2-[[(4-methoxyphenyl)diphenylmethyl]amino]-6-(phenylmethoxy)-9H-purin-9-yl]-2-(benzyloxymethyl)cyclopentanol-->(1S,2S,3S,5S)-5-(2-Amino-6-(benzyloxy)-9H-purin-9-yl)-3-(benzyloxy)-2-(benzyloxymethyl)cyclopentanol-->Benzyl chloromethyl ether-->Dibromomethane | | Preparation Products | 2-Amino-1,9-dihydro-9-[(1S,3R,4S)-4-(benzyloxy)-3-(benzyloxymethyl)-2-methylenecyclopentyl]-6H-purin-6-one |
|