- 2,6-Dihydroxypurine
-

- $9.90 / 1KG
-
2026-03-20
- CAS:69-89-6
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 5tons
- Xanthine
-

- $0.00 / 1KG
-
2025-12-12
- CAS:69-89-6
- Min. Order: 1KG
- Purity: 98%
- Supply Ability: 10MT
|
| | Xanthine Basic information |
| Product Name: | Xanthine | | Synonyms: | 2,6-DIOXOPURINE;2,6-DIOXYPURINE;9H-PURINE-2,6-DIOL;PURINE-2,6(1H,3H)-DIONE;TIMTEC-BB SBB004054;UREOUS ACID;XANTHINE;3,7-Dihydro-1H-purine-2,6-dione | | CAS: | 69-89-6 | | MF: | C5H4N4O2 | | MW: | 152.11 | | EINECS: | 200-718-6 | | Product Categories: | Heterocycles, Metabolites & Impurities;co-factor/nucleoside;Biochemistry;Nucleobases and their analogs;Nucleosides, Nucleotides & Related Reagents;Nucleic acids;Food Additives;Heterocycles;Metabolites & Impurities;Purines;Purine;Pyridines, Pyrimidines, Purines and Pteredines;bc0001;69-89-6 | | Mol File: | 69-89-6.mol | 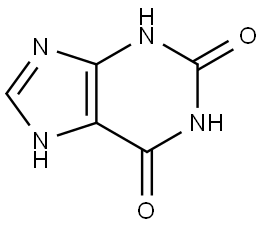 |
| | Xanthine Chemical Properties |
| Melting point | 300 °C | | Boiling point | 274.55°C (rough estimate) | | density | 1.5452 (rough estimate) | | vapor density | 5.3 (vs air) | | refractive index | 1.8500 (estimate) | | storage temp. | Keep in dark place,Sealed in dry,Room Temperature | | solubility | NH4OH: freely soluble | | pka | pKa 9.95 (Uncertain) | | form | Powder | | color | White to slightly yellow | | Odor | Odorless | | biological source | synthetic (organic) | | Water Solubility | Soluble in water(0.067g/L). | | Merck | 14,10059 | | BRN | 8733 | | Cosmetics Ingredients Functions | SKIN CONDITIONING | | InChI | 1S/C5H4N4O2/c10-4-2-3(7-1-6-2)8-5(11)9-4/h1H,(H3,6,7,8,9,10,11) | | InChIKey | LRFVTYWOQMYALW-UHFFFAOYSA-N | | SMILES | O=C1NC(=O)c2nc[nH]c2N1 | | LogP | -0.730 | | CAS DataBase Reference | 69-89-6(CAS DataBase Reference) | | NIST Chemistry Reference | Xanthine(69-89-6) | | EPA Substance Registry System | Xanthine (69-89-6) |
| Hazard Codes | Xi | | Risk Statements | 36-43-36/37/38 | | Safety Statements | 36/37-37/39-26 | | WGK Germany | 3 | | RTECS | ZD7700000 | | TSCA | TSCA listed | | HS Code | 29335990 | | Storage Class | 11 - Combustible Solids |
| | Xanthine Usage And Synthesis |
| Chemical Properties | White to off-white crystalline powder | | Uses | 2,6-Dihydroxypurine was fund in animal organs, yeast, patatoes, coffee beans, tea. 2,6-Dihydroxypurine can be used to relax and widen certain breathing passages of the lungs. It is also found that a large number of derivatives have adenoside receptor antagonist properties. | | Uses | Xanthine and xanthine oxidase system can be used to produce superoxide radicals. | | Definition | A poisonous colorless
crystalline organic compound that occurs
in blood, coffee beans, potatoes, and urine.
It is used as a chemical intermediate. | | Definition | Xanthine: a purine base, C5H4N4O2,found in many organisms. | | Origin | Xanthines (1H-purine-2,6(3H,7H)-diones) are purine based natural heterocyclic al�kaloids. They were first discovered in 1817 by German chemist Emil Fisher and later the name ‘xanthine’ was coined in 1899[1]. | | Biological Functions | In fact, xanthine and its derivatives act as intermediate molecules in generation of GMP, GDP and GTP via the salvage pathway inside the cells. In addition, xanthine plays imper�ative role in catabolism of nucleotides and nucleic acids as xanthine acts as precursor of uric acid. Therefore, xanthine contains similar skeleton as that of purines which forms the building blocks of unit of life i.e. ribonucleotides (RNA) and deoxyribo�nucleotides (DNA). The structural resemblance with two of the important purine de�rivatives Adenine and Guanine; makes xanthine a good therapeutic molecule[1].
| | Synthesis Reference(s) | Chemical and Pharmaceutical Bulletin, 22, p. 1200, 1974 DOI: 10.1248/cpb.22.1200 | | General Description | Xanthine is a purine that can be produced in the purine metabolic pathway via different precursors:
- Guanine deamination by guanine deaminase
- Hypoxanthine conversion by xanthine oxidoreductase
| | Biochem/physiol Actions | A high level of xanthine is implicated in metabolic disorders like Lesch-Nyhan syndrome. A xanthine-based biosensor may be useful for detecting xanthine in food and clinical samples. Blood and urine samples of patients with renal failure, gout and xanthinuria show high levels of xanthine. | | Synthesis | 1. Guanine reacts with sodium nitrite in hydrochloric acid to give intermediate AH01.
2. AH01 is hydrolyzed to yield crude xanthine.
3. The crude product is recrystallized twice from hydrochloric acid to increase purity (from 85% to 98% by HPLC).
4. The purified wet product is treated with ammonia to pH 7-8. The precipitated solid is filtered, washed with water, and vacuum-dried. | | Purification Methods | The monohydrate separates in a microcrystalline form on slow acidification with acetic acid of a solution of xanthine in dilute NaOH. It is also precipitated by addition of conc NH3 to its solution in hot 2N HCl (charcoal). After washing with H2O and EtOH, it is dehydrated by heating above 125o. Its solubility in H2O is 1 in 14,000parts at 16o and 1 in 1,500parts of boiling H2O, and separates as plates . It has no m, but the perchlorate has m 262-264o [Lister Purines Part II, Fused Pyrimidines Brown Ed, Wiley-Interscience pp252-253 1971, ISBN 0-471-38205-1]. [Beilstein 26 H 447, 26 I 131, 26 II 260, 26 III/IV 2327.] | | References | [1] Ahlawat, Jyoti Minakshi Sharma and Chandra S. Pundir. “Advances in xanthine biosensors and sensors: A review.” Enzyme and Microbial Technology 69 1 (2023). |
| | Xanthine Preparation Products And Raw materials |
|