|
|
| | TRANS-N-BOC-1,4-CYCLOHEXANEDIAMINE Basic information |
| Product Name: | TRANS-N-BOC-1,4-CYCLOHEXANEDIAMINE | | Synonyms: | N-Boc-transcyclohexanediamine;tert-butyl (1r,4r)-4-aminocyclohexylcarbamate;tert-Butyl (trans-4-aminocyclohex-1-yl)carbamate, trans-1,4-Diaminocyclohexane, N-BOC protected;trans-Cyclohexane-1,4-diamine, N-BOC protected;rel-tert-butyl N-[(1R,4R)-4-aMinocyclohexyl]carbaMate;trans-4-(Boc-aMino)cyclohexylaMine;Trans-1-(N-Boc AMino)-4-AMino cyclohexane;tert-butyl ((1r,4r)-4-aMinocyclohexyl)carbaMate hydrochloride | | CAS: | 177906-48-8 | | MF: | C11H22N2O2 | | MW: | 214.3 | | EINECS: | 618-108-7 | | Product Categories: | pharmacetical;177906-48-8 | | Mol File: | 177906-48-8.mol | 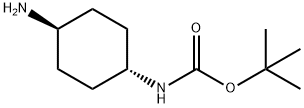 |
| | TRANS-N-BOC-1,4-CYCLOHEXANEDIAMINE Chemical Properties |
| Boiling point | 322.1±31.0 °C(Predicted) | | density | 1.02±0.1 g/cm3(Predicted) | | storage temp. | under inert gas (nitrogen or Argon) at 2–8 °C | | form | solid | | pka | 12.44±0.40(Predicted) | | color | White | | Water Solubility | Slightly soluble in water. | | InChI | InChI=1S/C11H22N2O2/c1-11(2,3)15-10(14)13-9-6-4-8(12)5-7-9/h8-9H,4-7,12H2,1-3H3,(H,13,14)/t8-,9- | | InChIKey | FEYLUKDSKVSMSZ-KYZUINATSA-N | | SMILES | C(OC(C)(C)C)(=O)N[C@@H]1CC[C@@H](N)CC1 | | CAS DataBase Reference | 177906-48-8(CAS DataBase Reference) |
| | TRANS-N-BOC-1,4-CYCLOHEXANEDIAMINE Usage And Synthesis |
| Uses | trans-N-Boc-1,4-cyclohexanediamine is used in the preparation of orally bioavailable and selective V1A receptor antagonist. | | Uses | trans-4-(Boc-amino)cyclohexylamine is used in the preparation of orally bioavailable and selective V1A receptor antagonist. | | Synthesis | Example 17. General procedure for the synthesis of tert-butyl trans-(4-aminocyclohexyl)carbamate (SB1-E-21-1) from di-tert-butyl dicarbonate and trans-1,4-cyclohexanediamine: To a methanol (100 mL) solution of SM-21-1 (2.0 g, 17.5 mmol) was slowly added dropwise a methanol (60 mL) solution of di-tert-butyl dicarbonate (1.1 g, 5.04 mmol). ) solution in methanol (60 mL) for 30 minutes. The reaction mixture was stirred at room temperature overnight. Upon completion of the reaction, the solvent was removed by concentration under reduced pressure. Water (50 mL) was added to the residue and stirring was continued for 20 minutes at room temperature, followed by filtration. The filtrate was extracted with ethyl acetate (120 mL x 2), and the organic phases were combined and washed sequentially with water (50 mL x 2) and saturated saline (50 mL x 2). The organic phase was dried with anhydrous sodium sulfate, filtered, and concentrated under reduced pressure to remove the solvent to afford the target product SB1-E-21-1 (off-white solid, 900 mg, 83% yield). | | References | [1] Chemistry - An Asian Journal, 2010, vol. 5, # 4, p. 877 - 886
[2] Patent: WO2016/160617, 2016, A2. Location in patent: Paragraph 00401
[3] Journal of Organic Chemistry, 1996, vol. 61, # 25, p. 8811 - 8818
[4] Patent: WO2016/116563, 2016, A1. Location in patent: Page/Page column 34-35
[5] Patent: WO2008/57468, 2008, A1. Location in patent: Page/Page column 352 |
| | TRANS-N-BOC-1,4-CYCLOHEXANEDIAMINE Preparation Products And Raw materials |
|